1911 Encyclopædia Britannica/Arachnida
ARACHNIDA, the zoological name given in 1815 by Lamarck (Gr. ἁράχνη, a spider) to a class which he instituted for the reception of the spiders, scorpions and mites, previously classified by Linnaeus in the order Aptera of his great group Insecta. Lamarck at the same time founded the class Crustacea for the lobsters, crabs and water-fleas, also until then included in the order Aptera of Linnaeus. Lamarck included the Thysanura and the Myriapoda in his class Arachnida. The Insecta of Linnaeus was a group exactly equivalent to the Arthropoda founded a hundred years later by Siebold and Stannius. It was thus reduced by Lamarck in area, and made to comprise only the six-legged, wing-bearing “Insecta.” For these Lamarck proposed the name Hexapoda; but that name has been little used, and they have retained to this day the title of the much larger Linnaean group, viz. Insecta. The position of the Arachnida in the great sub-phylum Arthropoda, according to recent anatomical and embryological researches, is explained in the article Arthropoda. The Arachnida form a distinct class or line of descent in the grade Euarthropoda, diverging (perhaps in common at the start with the Crustacea) from primitive Euarthropods, which gave rise also to the separate lines of descent known as the classes Diplopoda, Crustacea, Chilopoda and Hexapoda.
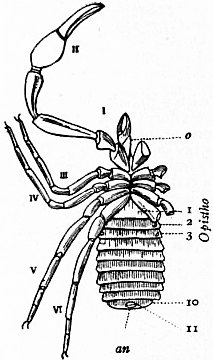
Limulus an Arachnid.—Modern views as to the classification and affinities of the Arachnida have been determined by the demonstration that Limulus and the extinct Eurypterines (Pterygotus, &c.) are Arachnida; that is to say, are identical in the structure and relation of so many important parts with Scorpio, whilst differing in those respects from other Arthropoda, that it is impossible to suppose that the identity is due to homoplasy or convergence, and the conclusion must be accepted that the resemblances arise from close genetic relationship. The view that Limulus, the king-crab, is an Arachnid was maintained as long ago as 1829 by Strauss-Dürckheim (1), on the ground of its possession of an internal cartilaginous sternum—also possessed by the Arachnida (see figs. 1, 2, 3, 4, 5 and 6)—and of the similarity of the disposition of the six leg-like appendages around the mouth in the two cases (see figs. 45 and 63). The evidence of the exact equivalence of the segmentation and appendages of Limulus and Scorpio, and of a number of remarkable points of agreement in structure, was furnished by Ray Lankester in an article published in 1881 (“Limulus an Arachnid,” Quart. Journ. Micr. Sci. vol. xxi. N.S.), and in a series of subsequent memoirs, in which the structure of the entosternum, of the coxal glands, of the eyes, of the veno-pericardiac muscles, of the respiratory lamellae, and of other parts, was for the first time described, and in which the new facts discovered were shown uniformly to support the hypothesis that Limulus is an Arachnid. A list of these memoirs is given at the close of this article (2, 3, 4, 5 and 13). The Eurypterines (Gigantostraca) were included in the identification, although at that time they were supposed to possess only five pairs of anterior or prosomatic appendages. They have now been shown to possess six pairs (fig. 47), as do Limulus and Scorpio.
The various comparisons previously made between the structure of Limulus and the Eurypterines on the one hand, and that of a typical Arachnid, such as Scorpio, on the other, had been vitiated by erroneous notions as to the origin of the nerves supplying the anterior appendages of Limulus (which were finally removed by Alphonse Milne-Edwards in his beautiful memoir (6) on the structure of that animal), and secondly by the erroneous identification of the double sternal plates of Limulus, called “chilaria,” by Owen, with a pair of appendages (7). Once the identity of the chilaria with the pentagonal sternal plate of the scorpion is recognized—an identification first insisted on by Lankester—the whole series of segments and appendages in the two animals, Limulus and Scorpio, are seen to correspond most closely, segment for segment, with one another (see figs. 7 and 8). The structure of the prosomatic appendages or legs is also seen to present many significant points of agreement (see figures), but a curious discrepancy existed in the six-jointed structure of the limb in Limulus, which differed from the seven-jointed limb of Scorpio by the defect of one joint. R. I. Pocock of the British Museum has observed that in Limulus a marking exists on the fourth joint, which apparently indicates a previous division of this segment into two, and thus establishes the agreement of Limulus and Scorpio in this small feature of the number of segments in the legs (see fig. 11).
It is not desirable to occupy the limited space of this article by a full description of the limbs and segments of Limulus and Scorpio. The reader is referred to the complete series of figures here given, with their explanatory legends (figs 12, 13, 14, 15). Certain matters, however, require comment and explanation to render the comparison intelligible. The tergites, or chitinized dorsal halves of the body rings, are fused to form a “prosomatic carapace,” or carapace of the prosoma, in both Limulus and Scorpio (see figs. 7 and 8). This region corresponds in both cases to six somites, as indicated by the presence of six pairs of limbs. On the surface of the carapace there are in both animals a pair of central eyes with simple lens and a pair of lateral eye-tracts, which in Limulus consist of closely-aggregated simple eyes, forming a “compound” eye, whilst in Scorpio they present several separate small eyes. The microscopic structure of the central and the lateral eyes has been shown by Lankester and A. G. Bourne (5) to differ; but the lateral eyes of Scorpio were shown by them to be similar in structure to the lateral eyes of Limulus, and the central eyes of Scorpio to be identical in structure with the central eyes of Limulus (see below).
 |
| Fig. 4.—Ventral surface of the same entosternum as that drawn in fig. 3. Letters as in fig. 3 with the addition of NC, neural canal or foramen. (After Lankester, loc. cit.) |
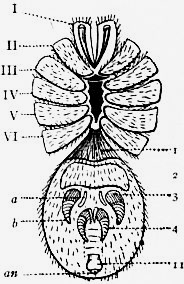
Following the prosoma is a region consisting of six segments (figs. 14 and 15), each carrying a pair of plate-like appendages in both Limulus and Scorpio. This region is called the mesosoma. The tergites of this region and those of the following region, the metasoma, are fused to form a second or posterior carapace in Limulus, whilst remaining free in Scorpio. The first pair of foliaceous appendages in each animal is the genital operculum; beneath it are found the openings of the genital ducts. The second pair of mesosomatic appendages in Scorpio are known as the “pectens.” Each consists of an axis, bearing numerous blunt tooth-like processes arranged in a series. This is represented in Limulus by the first gill-bearing appendage. The leaves (some 150 in number) of the gill-book (see figure) correspond to the tooth-like processes of the pectens of Scorpio. The next four pairs of appendages (completing the mesosomatic series of six) consist, in both Scorpio and Limulus, of a base carrying each 130 to 150 blood-holding, leaf-like plates, lying on one another like the leaves of a book. Their minute structure is closely similar in the two cases; the leaf-like plates receive blood from the great sternal sinus, and serve as respiratory organs. The difference between the gill-books of Limulus and the lung-books of Scorpio depends on the fact that the latter are adapted to aerial respiration, while the former serve for aquatic respiration. The appendage carrying the gill-book stands out on the surface of the body in Limulus, and has other portions developed besides the gill-book and its base; it is fused with its fellow of the opposite side. On the other hand, in Scorpio, the gill-book-bearing appendage has sunk below the surface, forming a recess or chamber for itself, which communicates with the exterior by an oval or circular “stigma” (fig. 10, stg). That this in-sinking has taken place, and that the lung-books or in-sunken gill-books of Scorpio really represent appendages (that is to say, limbs or parapodia) is proved by their developmental history (see figs. 17 and 18). They appear at first as outstanding processes on the surface of the body.
The exact mode in which the in-sinking of superficial outstanding limbs, carrying gill-lamellae, has historically taken place has been a matter of much speculation. It was to be hoped that the specimen of the Silurian scorpion (Palaeophonus) from Scotland, showing the ventral surface of the mesosoma (fig. 49), would throw light on this matter; but the specimen recently carefully studied by the writer and Pocock reveals neither gill-bearing limbs nor stigmata. The probability appears to be against an actual introversion of the appendage and its lamellae, as was at one time suggested by Lankester. It is probable that such an in-sinking as is shown in the accompanying diagram has taken place (fig. 15); but we are yet in need of evidence as to the exact equivalence of margins, axis, &c., obtaining between the lung-book of Scorpio and the gill-book of Limulus. Zoologists are familiar with many instances (fishes, crustaceans) in which the protective walls of a water-breathing organ or gill-apparatus become converted into an air-breathing organ or lung, but there is no other case known of the conversion of gill processes themselves into air-breathing plates.
| [According to the system of numbering explained in the text, if VII is the tergum of the |
| praegenital somite (as is probable) it should be labelled Prg without any number, and the |
| somites VIII to XIII should be lettered 1 to 6, indicating that they are the six normal somites |
| of the mesosoma; whilst XV to XVIII should be replaced by the numbers 7 to 12—an |
| additional suppressed segment (making up the typical six) being reckoned to the |
| metasomatic fusion.] |
(From Lankester, Q. J. Micr. Sci. vol. xxi., 1881.) |
The identification of the lung-books of Scorpio with the gill-books of Limulus is practically settled by the existence of the pectens in Scorpio (fig. 14, VIII) on the second mesosomatic somite. There is no doubt that these are parapodial or limb appendages, carrying numerous imbricated secondary processes, and therefore comparable in essential structure to the leaf-bearing plates of the second mesosomatic somite of Limulus. They have remained unenclosed and projecting on the surface of the body, as once were the appendages of the four following somites. But they have lost their respiratory function. In non-aquatic life such an unprotected organ cannot subserve respiration. The “pectens” have become more firmly chitinized and probably somewhat altered in shape as compared with their condition in the aquatic ancestral scorpions. Their present function in scorpions is not ascertained. They are not specially sensitive under ordinary conditions, and may be touched or even pinched without causing any discomfort to the scorpion. It is probable that they acquire special sensibility at the breeding season and serve as “guides” in copulation. The shape of the legs and the absence of paired terminal claws in the Silurian Palaeophonus (see figs. 48 and 49) as compared with living scorpions (see fig. 10) show that the early scorpions were aquatic, and we may hope some day in better-preserved specimens than the two as yet discovered, to find the respiratory organs of those creatures in the condition of projecting appendages serving aquatic respiration somewhat as in Limulus, though not necessarily repeating the exact form of the broad plates of Limulus.
It is important to note that the series of lamellae of the lung-book and the gill-book correspond exactly in structure, the narrow, flat blood-space in the lamellae being interrupted by pillar-like junctions of the two surfaces in both cases (see Lankester (4)), and the free surfaces of the adjacent lamellae being covered with a very delicate chitinous cuticle which is drawn out into delicate hairs and processes. The elongated axis which opens at the stigma in Scorpio and which can be cleared of soft, surrounding tissues and coagulated blood so as to present the appearance of a limb axis carrying the book-like leaves of the lung is not really, as it would seem to be at first sight, the limb axis. That is necessarily a blood-holding structure and is obliterated and fused with soft tissues of the sternal region so that the lamellae cannot be detached and presented as standing out from it. The apparent axis or basal support of the scorpion’s lung-books shown in the figures, is a false or secondary axis and merely a part of the infolded surface which forms the air-chamber. The maceration of the soft parts of a scorpion preserved in weak spirit and the cleaning of the chitinized in-grown cuticle give rise to the false appearance of a limb axis carrying the lamellae. The margins of the lamellae of the scorpion’s lung-book, which are lowermost in the figures (fig. 15) and appear to be free, are really those which are attached to the blood-holding axis. The true free ends are those nearest the stigma.
Passing on now from the mesosoma we come in Scorpio to the metasoma of six segments, the first of which is broad whilst the rest are cylindrical. The last is perforated by the anus and carries the post-anal spine or sting. The somites of the metasoma carry no parapodia. In Limulus the metasoma is practically suppressed. In the allied extinct Eurypterines it is well developed, and resembles that of Scorpio. In the embryo Limulus (fig. 42) the six somites of the mesosoma are not fused to form a carapace at an early stage, and they are followed by three separately marked metasomatic somites; the other three somites of the metasoma have disappeared in Limulus, but are represented by the unsegmented prae-anal region. It is probable that we have in the metasoma of Limulus a case of the disappearance of once clearly demarcated somites. It would be possible to suppose, on the other hand, that new somites are only beginning to make their appearance here. The balance of various considerations is against the latter hypothesis. Following the metasoma in Limulus, we have as in Scorpio the post-anal spine—in this case not a sting, but a powerful and important organ of locomotion, serving to turn the animal over when it has fallen upon its back. The nature of the post-anal spine has been strangely misinterpreted by some writers. Owen (7) maintained that it represented a number of coalesced somites, regardless of its post-anal position and mode of development. The agreement of the grouping of the somites, of the form of the parapodia (appendages, limbs) in each region, of the position of the genital aperture and operculum, of the position and character of the eyes, and of the powerful post-anal spines not seen in other Arthropods, is very convincing as to the affinity of Limulus and Scorpio. Perhaps the most important general agreement of Scorpio compared with Limulus and the Eurypterines is the division of the body into the three regions (or tagmata)—prosoma, mesosoma and metasoma—each consisting of six segments, the prosoma having leg-like appendages, the mesosoma having foliaceous appendages, and the metasoma being destitute of appendages.
In 1893, some years after the identification of the somites of Limulus with those of Scorpio, thus indicated, had been published, zoologists were startled by the discovery by a Japanese zoologist, Kishinouye (8), of a seventh prosomatic somite in the embryo of Limulus longispina. This was seen in longitudinal sections, as shown in fig. 19. The simple identification of somite with somite in Limulus and Scorpio seemed to be threatened by this discovery. But in 1896 Dr August Brauer of Marburg (9) discovered in the embryo of Scorpio a seventh prosomatic somite (see VII PrG, figs. 17 and 18), or, if we please so to term it, a praegenital somite, hitherto unrecognized. In the case of Scorpio this segment is indicated in the embryo by the presence of a pair of rudimentary appendages, carried by a well-marked somite. As in Limulus, so in Scorpio, this unexpected somite and its appendages disappear in the course of development. In fact, more or less complete “excalation” of the somite takes place. Owing to its position it is convenient to term the somite which is excalated in Limulus and Scorpio “the praegenital somite.” It appears not improbable that the sternal plates wedged in between the last pair of legs in both Scorpio and Limulus, viz. the pentagonal sternite of Scorpio (fig. 10) and the chilaria of Limulus (see figs. 13 and 20), may in part represent in the adult the sternum of the excalated praegenital somite. This has not been demonstrated by an actual following out of the development, but the position of these pieces and the fact that they are (in Limulus) supplied by an independent segmental nerve, favours the view that they may comprise the sternal area of the vanished praegenital somite. This interpretation, however, of the “metasternites” of Limulus and Scorpio is opposed by the coexistence in Thelyphonus (figs. 55, 57 and 58) of a similar metasternite with a complete praegenital somite. H. J. Hansen (10) has recognized that the “praegenital somite” persists in a rudimentary condition, forming a “waist” to the series of somites in the Pedipalpi and Araneae. The present writer is of opinion that it will be found most convenient to treat this evanescent somite as something special, and not to attempt to reckon it to either the prosoma or the mesosoma. 
Fig. 11.—Third leg of Limulus polyphemus, showing the division of the fourth segment of the leg by a groove S into two, thus giving seven segments to the leg as in scorpion.
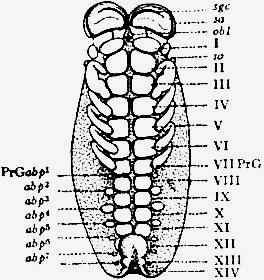
(From a drawing by Pocock.)These will then remain as typically composed each of six appendage-bearing somites—the prosoma comprising in addition the ocular prosthomere.[1] When the praegenital somite or traces of it are present it should not be called “the seventh prosomatic” or the “first mesosomatic,” but simply the “praegenital somite.” The first segment of the mesosoma of Scorpio and Limulus thus remains the first segment, and can be identified as such throughout the Eu-arachnida, carrying as it always does the genital apertures. But it is necessary to remember, in the light of recent discoveries, that the sixth prosomatic pair of appendages is carried on the seventh somite of the whole series, there being two prosthomeres or somites in front of the mouth, the first carrying the eyes, the second the chelicerae; also that the first mesosomatic or genital somite is not the seventh or even the eighth of the whole senes of somites which have been historically present, but is the ninth, owing to the presence or to the excalation of a praegenital somite. It seems that confusion and trouble will be best avoided by abstaining from the introduction of the non-evident somites, the ocular and the praegenital, into the numerical nomenclature of the component somites of the three great body regions. We shall, therefore, ignoring the ocular somite, speak of the first, second, third, fourth, fifth and sixth leg-bearing somites of the prosoma, and indicate the appendages by the Roman numerals, I, II, III, IV, V, VI, and whilst ignoring the praegenital somite we shall speak of the first, second, third, &c., somite of the mesosoma or opisthosoma (united mesosoma and metasoma) and indicate them by the Arabic numerals.
There are a number of other important points of structure besides those referring to the somites and appendages in which Limulus agrees with Scorpio or other Arachnida and differs from other Arthropoda. The chief of these are as follows:—
1. The Composition of the Head (that is to say, of the anterior part of the prosoma) with especial Reference to the Region in Front of the Mouth.—It appears (see Arthropoda) that there is embryological evidence of the existence of two somites in Arachnida which were originally post-oral, but have become prae-oral by adaptational shifting of the oral aperture. These forwardly-slipped somites are called “prosthomeres.” The first of these has, in Arachnids as in other Arthropods, its pair of appendages represented by the eyes. The second has for its pair of appendages the small pair of limbs which in all living Arachnids is either chelate or retrovert (as in spiders), and is known as the chelicerae. It is possible, as maintained by some writers (Patten and others), that the lobes of the cerebral nervous mass in Arachnids indicate a larger number of prosthomeres as having fused in this region, but there is no embryological evidence at present which justifies us in assuming the existence in Arachnids of more than two prosthomeres. The position of the chelicerae of Limulus and of the ganglionic nerve-masses from which they receive their nerve-supply, is closely similar to that of the same structures in Scorpio. The cerebral mass is in Limulus more easily separated by dissection as a median lobe distinct from the laterally-placed ganglia of the chelceral somite than is the case in Scorpio, but the relations are practically the same in the two forms. Formerly it was supposed that in Limulus both the chelicerae and the next following pair of appendages were prosthomerous, as in Crustacea, but the dissections of Alphonse Milne-Edwards (6) demonstrated the true limitations of the cerebrum, whilst embryological researches have done as much for Scorpio. Limulus thus agrees with Scorpio and differs from the Crustacea, in which there are three prosthomeres—one ocular and two carrying palpiform appendages. It is true that in the lower Crustacea (Apus, &c.) we have evidence of the gradual movement forward of the nerve-ganglia belonging to these palpiform appendages. But although in such lower Crustacea the nerve-ganglia of the third prosthomere have not fused with the anterior nerve-mass, there is no question as to the prae-oral position of two appendage-bearing somites in addition to the ocular prosthomere. The Crustacea have, in fact, three prosthomeres in the head and the Arachnida only two, and Limulus agrees with the Arachnida in this respect and differs from the Crustacea. The central nervous systems of Limulus and of Scorpio present closer agreement in structure than can be found when a Crustacean is compared with either. The wide divarication of the lateral cords in the prosoma and their connexion by transverse commissures, together with the “attraction” of ganglia to the prosomatic ganglion group which properly belong to hinder segments, are very nearly identical in the two animals. The form and disposition of the ganglion cells are also peculiar and closely similar in the two. (See Patten (42) for important observations on the neuromeres, &c., of Limulus and Scorpio.)
(From Lankester, loc. cit.)
2. The Minute Structure of the Central Eyes and of the Lateral Eyes.—Limulus agrees with Scorpio not only in having a pair of central eyes and also lateral eyes, but in the microscopic structure of those organs, which differs in the central and lateral eyes respectively. The central eyes are “simple eyes,” that is to say, have a single lens, and are hence called “monomeniscous.” The lateral eyes are in Limulus “compound eyes,” that is to say, consist of many lenses placed close together; beneath each lens is a complex of protoplasmic cells, in which the optic nerve terminates. Each such unit is termed an “ommatidium.” The lateral eyes of Scorpio consist of groups of separate small lenses each with its ommatidium, but they do not form a continuous compound eye as in Limulus. The ommatidium (soft structure beneath the lens-unit of a compound eye) is very simple in both Scorpio and Limulus. It consists of a single layer of cells, continuous with those which secrete the general chitinous covering of the prosoma. The cells of the ommatidium are a good deal larger than the neighbouring common cells of the epidermis. They secrete the knob-like lens (fig. 22). But they also receive the nerve fibres of the optic nerve. They are at the same time both optic nerve-end cells, that is to say, retina cells, and corneagen cells or secretors of the chitinous lens-like cornea. In Limulus (fig. 23) each ommatidium has a peculiar ganglion cell developed in a central position, whilst the ommatidium of the lateral eyelets of Scorpio shows small intermediate cells between the larger nerve-end cells. The structure of the lateral eye of Limulus was first described by Grenacher, and further and more accurately by Lankester and Bourne (5) and by Watase; that of Scorpio by Lankester and Bourne, who showed that the statements of von Graber were erroneous, and that the lateral eyes of Scorpio have a single cell-layered or “monostichous” ommatidium like that of Limulus. Watase has shown, in a very convincing way, how by deepening the pit-like set of cells beneath a simple lens the more complex ommatidia of the compound eyes of Crustacea and Hexapoda may be derived from such a condition as that presented in the lateral eyes of Limulus and Scorpio. (For details the reader is referred to Watase (11) and to Lankester and Bourne (5).) The structure of the central eyes of Scorpio and spiders and also of Limulus differs essentially from that of the lateral eyes in having two layers of cells (hence called diplostichous) beneath the lens, separated from one another by a membrane (figs. 24 and 25). The upper layer is the corneagen and secretes the lens, the lower is the retinal layer. The mass of soft cell-structures beneath a large lens of a central eye is called an “ommatoeum.” It shows in Scorpio and Limulus a tendency to segregate into minor groups or “ommatidia.” It is found that in embryological growth the retinal layer of the central eyes forms as a separate pouch, which is pushed in laterally beneath the corneagen layer from the epidermic cell layer. Hence it is in origin double, and consists of a true retinal layer and a post-retinal layer (fig. 24, B), though these are not separated by a membrane. Accordingly the diplostichous ommatoeum or soft tissue of the Arachnid’s central eye should strictly be called “triplostichous,” since the deep layer is itself doubled or folded. The retinal cells of both the lateral and central eyes of Limulus and Scorpio produce cuticular structures on their sides; each such piece is a rhabdomere and a number (five or ten) uniting form a rhabdom (fig. 26). In the specialized ommatidia of the compound eyes of Crustacea and Hexapods the rhabdom is an important structure.[2] It is a very significant fact that the lateral and central eyes of Limulus and Scorpio not only agree each with each in regard to their monostichous and diplostichous structure, but also in the formation in both classes of eyes of rhabdomeres and rhabdoms in which the component pieces are five or a multiple of five (fig. 26). Whilst each unit of the lateral eye of Limulus has a rhabdom of ten[3] pieces forming a star-like chitinous centre in section, each lateral eye of Scorpio has several rhabdoms of five or less rhabdomeres, indicating that the Limulus lateral eye-unit is more specialized than the detached lateral eyelet of Scorpio, so as to present a coincidence of one lens with one rhabdom. Numerous rhabdomeres (grouped as rhabdoms in Limulus) are found in the retinal layer of the central eyes also.
|
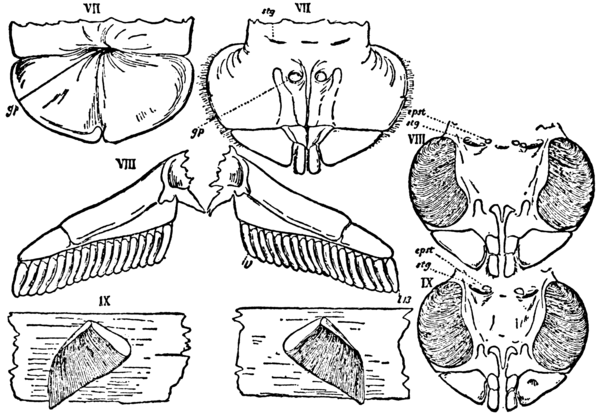
Fig. 14.—The first three pairs of mesosomatic appendages of Scorpio and Limulus compared. |
| VII, The genital operculum. | gp, Genital pore. |
| VIII, The pectens of Scorpio and the first | epst, Epistigmatic sclerite. |
| branchial plate of Limulus. | stg, Stigma or orifice of the hollow tendons of the |
| IX, The first pair of lung-books of Scorpio and the | branchial plates of Limulus. |
| second branchial plate of Limulus. |
(After Lankester, loc. cit.)
Whilst Limulus agrees thus closely with Scorpio in regard to the eyes, it is to be noted that no Crustacean has structures corresponding to the peculiar diplostichous central eyes, though these occur again (with differences in detail) in Hexapoda. Possibly, however, an investigation of the development of the median eyes of some Crustacea (Apus, Palaemon) may prove them to be diplostichous in origin.
3. The so-called “Coxal Glands.”—In 1882 (Proc. Roy. Soc. No. 221) Lankester described under the name “coxal glands” a pair of brilliantly white oviform bodies lying in the Scorpion’s prosoma immediately above the coxae of the fifth and sixth pairs of legs (fig. 27). These bodies had been erroneously supposed by Newport (12) and other observers to be glandular outgrowths of the alimentary canal. They are really excretory glands, and communicate with the exterior by a very minute aperture on the posterior face of the coxa of the fifth limb on each side. When examined with the microscope, by means of the usual section method, they are seen to consist of a labyrinthine tube lined with peculiar cells, each cell having a deep vertically striated border on the surface farthest from the lumen, as is seen in the cells of some renal organs. The coils and branches of the tube are packed by connective tissue and blood spaces. A similar pair of coxal glands, lobate instead of ovoid in shape, was described by Lankester in Mygale, and it was also shown by him that the structures in Limulus called “brick-red glands” by Packard have the same structure and position as the coxal glands of Scorpio and Mygale. In Limulus these organs consist each of four horizontal lobes lying on the coxal margin of the second, third, fourth, and fifth prosomatic limbs, the four lobes being connected to one another by a transverse piece or stem (fig. 28). Microscopically their structure is the same in essentials as that of the coxal glands of Scorpio (13). Coxal glands have since been recognized and described in other Arachnida. In 1900 it was shown that the coxal gland of Limulus is provided with a very delicate thin-walled coiled duct which opens, even in the adult condition, by a minute pore on the coxa of the fifth leg (Patten and Hazen, 13a). Previously to this, Lankester’s pupil Gulland had shown (1885) that in the embryo the coxal gland is a comparatively simple tube, which opens to the exterior in this position and by its other extremity into a coelomic space. Similar observations were made by Laurie (17) in Lankester’s laboratory (1890) with regard to the early condition of the coxal gland of Scorpio, and by Bertkau (41) as to that of the spider Atypus. H. M. Bernard (13b) showed that the opening remains in the adult scorpion. In all the embryonic or permanent opening is on the coxa of the fifth pair of prosomatic limbs. Thus an organ newly discovered in Scorpio was found to have its counterpart in Limulus.
The name “coxal gland” needs to be carefully distinguished from “crural gland,” with which it is apt to be confused. The crural glands, which occur in many terrestrial Arthropods, are epidermal in origin and totally distinct from the coxal glands. The coxal glands of the Arachnida are structures of the same nature as the green glands of the higher Crustacea and the so-called “shell glands” of the Entomostraca. The latter open at the base of the fifth pair of limbs of the Crustacean, just as the coxal glands open on the coxal joint of the fifth pair of limbs of the Arachnid. Both belong to the category of “coelomoducts,” namely, tubular or funnel-like portions of the coelom opening to the exterior in pairs in each somite (potentially,) and usually persisting in only a few somites as either “urocoels” (renal organs) or “gonocoels” (genital tubes). In Peripatus they occur in every somite of the body. They have till recently been very generally identified with the nephridia of Chaetopod worms, but there is good reason for considering the true nephridia (typified by the nephridia of the earthworm) as a distinct class of organs (see Lankester in vol. ii. chap. iii. of A Treatise on Zoology, 1900). The genital ducts of Arthropoda are, like the green glands, shell glands and coxal glands, to be regarded as coelomoducts (gonocoels). The coxal glands do not establish any special connexion between Limulus and Scorpio, since they also occur in the same somite in the lower Crustacea, but it is to be noted that the coxal glands of Limulus are in minute structure and probably in function more like those of Arachnids than those of Crustacea.
4. The Entosternites and their Minute Structure.—Strauss-Dürckheim (1) was the first to insist on the affinity between Limulus and the Arachnids, indicated by the presence of a free suspended entosternum or plastron or entosternite in both. We have figured here (figs. 1 to 6) the entosternites of Limulus, Scorpio and Mygale. Lankester some years ago made a special study of the histology (3) of these entosternites for the purpose of comparison, and also ascertained the relations of the very numerous muscles which are inserted into them (4). The entosternites are cartilaginous in texture, but they have neither the chemical character nor the microscopic structure of the hyaline cartilage of Vertebrates. They yield chitin in place of chondrin or gelatin—as does also the cartilage of the Cephalopod’s endoskeleton. In microscopic structure they all present the closest agreement with one another. We find a firm, homogeneous or sparsely fibrillated matrix in which are embedded nucleated cells (corpuscles of protoplasm) arranged in rows of three, six or eight, parallel with the adjacent lines of fibrillation.
A minute entosternite having the above-described structure is found in the Crustacean Apus between the bases of the mandibles, and also in the Decapoda in a similar position, but in no Crustacean does it attain to any size or importance. On the other hand, the entosternite of the Arachnida is a very large and important feature in the structure of the prosoma, and must play an important part in the economy of these organisms. In Limulus (figs. 1 and 2) it has as many as twenty-five pairs of muscles attached to it, coming to it from the bases of the surrounding limbs and from the dorsal carapace and from the pharynx. It consists of an oblong plate 2 in. in length and 1 in breadth, with a pair of tendinous outgrowths standing out from it at right angles on each side. It “floats” between the prosomatic nerve centres and the alimentary canal. In each somite of the mesosoma is a small, free entosternite having a similar position, but below or ventral to the nerve cords, and having a smaller number of muscles attached to it. The entosternite was probably in origin part of the fibrous connective tissue lying close to the integument of the sternal surface—giving attachment to muscles corresponding more or less to those at present attached to it. It became isolated and detached, why or with what advantage to the organism it is difficult to say, and at that period of Arachnidan development the great ventral nerve cords occupied a more lateral position than they do at present. We know that such a lateral position of the nerve cords preceded the median position in both Arthropoda and Chaetopoda. Subsequently to the floating off of the entosternite the approximation of the nerve cords took place in the prosoma, and thus they were able to take up a position below the entosternite. In the mesosoma the approximation had occurred before the entosternites were formed.
In the scorpion (figs. 3 and 4) the entosternite has tough membrane-like outgrowths which connect it with the body-wall, both dorsally and ventrally forming an oblique diaphragm, cutting off the cavity of the prosoma from that of the mesosoma. It was described by Newport as “the diaphragm.” Only the central and horizontal parts of this structure correspond precisely to the entosternite of Limulus: the right and left anterior processes (marked ap in figs. 3 and 4, and RAP, LAP, in figs. 1 and 2) correspond in the two animals, and the median lateral process lmp of the scorpion represents the tendinous outgrowths ALR, PLR of Limulus. The scorpion’s entosternite gives rise to outgrowths, besides the great posterior flaps, pf, which form the diaphragm, unrepresented in Limulus. These are a ventral arch forming a neural canal through which the great nerve cords pass (figs. 3 and 4, snp), and further a dorsal gastric canal and arterial canal which transmit the alimentary tract and the dorsal artery respectively (figs. 3 and 4, GC, DR).
In Limulus small entosternites are found in each somite of the appendage-bearing mesosoma, and we find in Scorpio, in the only somite of the mesosoma which has a well-developed pair of appendages, that of the pectens, a small entosternite with ten pairs of muscles inserted into it. The supra-pectinal entosternite lies ventral to the nerve cords.
In Mygale (figs. 5 and 6) the form of the entosternite is more like that of Limulus than is that of Scorpio. The anterior notch Ph.N. is similar to that in Limulus, whilst the imbricate triangular pieces of the posterior median region resemble the similarly-placed structures of Limulus in a striking manner.
It must be confessed that we are singularly ignorant as to the functional significance of these remarkable organs—the entosternites. Their movement in an upward or downward direction in Limulus and Mygale must exert a pumping action on the blood contained in the dorsal arteries and the ventral veins respectively. In Scorpio the completion of the horizontal plate by oblique flaps, so as to form an actual diaphragm shutting off the cavity of the prosoma from the rest of the body, possibly gives to the organs contained in the anterior chamber a physiological advantage in respect of the supply of arterial blood and its separation from the venous blood of the mesosoma. Possibly the movement of the diaphragm may determine the passage of air into or out of the lung-sacs. Muscular fibres connected with the suctorial pharynx are in Limulus inserted into the entosternite, and the activity of the two organs may be correlated.
5. The Blood and the Blood-vascular System.—The blood fluids of Limulus and Scorpio are very similar. Not only are the blood corpuscles of Limulus more like in form and granulation to those of Scorpio than to those of any Crustacean, but the fluid is in both animals strongly impregnated with the blue-coloured respiratory proteid, haemocyanin. This body occurs also in the blood of Crustacea and of Molluscs, but its abundance in both Limulus and Scorpio is very marked, and gives to the freshly-shed blood a strong indigo-blue tint.
The great dorsal contractile vessel or “heart” of Limulus is closely similar to that of Scorpio; its ostia or incurrent orifices are placed in the same somites as those of Scorpio, but there is one additional posterior pair. The origin of the paired arteries from the heart differs in Limulus from the arrangement obtaining in Scorpio, in that a pair of lateral commissural arteries exist in Limulus (as described by Alphonse Milne-Edwards (6)) leading to a suppression of the more primitive direct connexion of the four pairs of posterior lateral arteries and of the great median posterior arteries with the heart itself (fig. 29).
The arterial system is very completely developed in both Limulus and Scorpio, branching repeatedly until minute arterioles are formed, not to be distinguished from true capillaries; these open into irregular swollen vessels which are the veins or venous sinuses. A very remarkable feature in Limulus, first described by Owen, is the close accompaniment of the prosomatic nerve centres and nerves by arteries, so close indeed that the great ganglion mass and its out-running nerves are actually sunk in or invested by arteries. The connexion is not so intimate in Scorpio, but is nevertheless a very close one, closer than we find in any other Arthropods in which the arterial system is well developed, e.g. the Myriapoda and some of the arthrostracous Crustacea. It seems that there is a primitive tendency in the Arthropoda for the arteries to accompany the nerve cords, and a “supra-spinal” artery—that is to say, an artery in close relation to the ventral nerve cords—has been described in several cases. On the other hand, in many Arthropods, especially those which possess tracheae, the arteries do not have a long course, but soon open into wide blood sinuses. Scorpio certainly comes nearer to Limulus in the high development of its arterial system, and the intimate relation of the anterior aorta and its branches to the nerve centres and great nerves, than does any other Arthropod.
An arrangement of great functional importance in regard to the venous system must now be described, which was shown in 1883 by Lankester to be common to Limulus and Scorpio. This arrangement has not hitherto been detected in any other class than the Arachnida, and if it should ultimately prove to be peculiar to that group, would have considerable weight as a proof of the close genetic affinity of Limulus and Scorpio.
The great pericardial sinus is strongly developed in both animals. Its walls are fibrous and complete, and it holds a considerable volume of blood when the heart itself is contracted. Opening in pairs in each somite, right and left into the pericardial sinus are large veins, which bring the blood respectively from the gill-books and the lung-books to that chamber, whence it passes by the ostia into the heart. The blood is brought to the respiratory organs in both cases by a great venous collecting sinus having a ventral median position. In both animals the wall of the pericardial sinus is connected by vertical muscular bands to the wall of the ventral venous sinus (its lateral expansions around the lung-books in Scorpio) in each somite through which the pericardium passes. There are seven pairs of these veno-pericardiac vertical muscles in Scorpio, and eight in Limulus (see figs. 30, 31, 32). It is obvious that the contraction of these muscles must cause a depression of the floor of the pericardium and a rising of the roof of the ventral blood sinus, and a consequent increase of volume and flow of blood to each. Whether the pericardium and the ventral sinus are made to expand simultaneously or all the movement is made by one only of the surfaces concerned, must depend on conditions of tension. In any case it is clear that we have in these muscles an apparatus for causing the blood to flow differentially in increased volume into either the pericardium, through the veins leading from the respiratory organs, or from the body generally into the great sinuses which bring the blood to the respiratory organs. These muscles act so as to pump the blood through the respiratory organs.
(After Lankester and Bourne, Q. J. Mic. Sci., 1883.)
It is not surprising that with so highly developed an arterial system Limulus and Scorpio should have a highly developed mechanism for determining the flow of blood to the respiratory organs. That this is, so to speak, a need of animals with localized respiratory organs is seen by the existence of provisions serving a similar purpose in other animals, e.g. the branchial hearts of the Cephalopoda.
The veno-pericardiac muscles of Scorpio were seen and figured by Newport but not described by him. Those of Limulus were described and figured by Alphonse Milne-Edwards, but he called them merely “transparent ligaments,” and did not discover their muscular structure. They are figured and their importance for the first time recognized in the memoir on the muscular and skeletal systems of Limulus and Scorpio by Lankester, Beck and Bourne (4).
6. Alimentary Canal and Gastric Glands.—The alimentary canal in Scorpio, as in Limulus, is provided with a powerful suctorial pharynx, in the working of which extrinsic muscles take a part. The mouth is relatively smaller in Scorpio than in Limulus—in fact is minute, as it is in all the terrestrial Arachnida which suck the juices of either animals or plants. In both, the alimentary canal takes a straight course from the pharynx (which bends under it downwards and backwards towards the mouth in Limulus) to the anus, and is a simple, narrow, cylindrical tube (fig. 33). The only point in which the gut of Limulus resembles that of Scorpio rather than that of any of the Crustacea, is in possessing more than a single pair of ducts or lateral outgrowths connected with ramified gastric glands or gastric caeca. Limulus has two pairs of these, Scorpio as many as six pairs. The Crustacea never have more than one pair. The minute microscopic structure of the gastric glands in the two animals is practically identical. The functions of these gastric diverticula have never been carefully investigated. It is very probable that in Scorpio they do not serve merely to secrete a digestive fluid (shown in other Arthropoda to resemble the pancreatic fluid), but that they also become distended by the juices of the prey sucked in by the scorpion—as certainly must occur in the case of the simple unbranched gastric caeca of the spiders.
The most important difference which exists between the structure of Limulus and that of Scorpio is found in the hinder region of the alimentary canal. Scorpio is here provided with a single or double pair of renal excretory tubes, which have been identified by earlier authors with the Malpighian tubes of the Hexapod and Myriapod insects. Limulus is devoid of any such tubes. We shall revert to this subject below.
(After Lankester.)
7. Ovaries and Spermaries: Gonocoels and Gonoducts.—The scorpion is remarkable for having the specialized portion of coelom from the walls of which egg-cells or sperm-cells are developed according to sex, in the form of a simple but extensive network. It is not a pair of simple tubes, nor of dendriform tubes, but a closed network. The same fact is true of Limulus, as was shown by Owen (7) in regard to the ovary, and by Benham (14) in regard to the testis. This is a very definite and remarkable agreement, since such a reticular gonocoel is not found in Crustacea (except in the male Apus). Moreover, there is a significant agreement in the character of the spermatozoa of Limulus and Scorpio. The Crustacea are—with the exception of the Cirrhipedia—remarkable for having stiff, motionless spermatozoids. In Limulus Lankester found (15) the spermatozoa to possess active flagelliform “tails,” and to resemble very closely those of Scorpio which, as are those of most terrestrial Arthropoda, are actively motile. This is a microscopic point of agreement, but is none the less significant.
In regard to the important structures concerned with the fertilization of the egg, Limulus and Scorpio differ entirely from one another. The eggs of Limulus are fertilized in the sea after they have been laid. Scorpio, being a terrestrial animal, fertilizes by copulation. The male possesses elaborate copulatory structures of a chitinous nature, and the eggs are fertilized in the female without even quitting the place where they are formed on the wall of the reticular gonocoel. The female scorpion is viviparous, and the young are produced in a highly developed condition as fully formed scorpions.
Differences between Limulus and Scorpio.—We have now passed in review the principal structural features in which Limulus agrees with Scorpio and differs from other Arthropoda. There remains for consideration the one important structural difference between the two animals. Limulus agrees with the majority of the Crustacea in being destitute of renal excretory caeca or tubes opening into the hinder part of the gut. Scorpio, on the other hand, in common with all air-breathing Arthropoda except Peripatus, possesses these tubules, which are often called Malpighian tubes. A great deal has been made of this difference by some writers. It has been considered by them as proving that Limulus, in spite of all its special agreements with Scorpio (which, however, have scarcely been appreciated by the writers in question), really belongs to the Crustacean line of descent, whilst Scorpio, by possessing Malpighian tubes, is declared to be unmistakably tied together with the other Arachnida to the tracheate Arthropods, the Hexapods, Diplopods, and Chilopods, which all possess Malpighian tubes.
It must be pointed out that the presence or absence of such renal excretory tubes opening into the intestine appears to be a question of adaptation to the changed physiological conditions of respiration, and not of morphological significance, since a pair of renal excretory tubes of this nature is found in certain Amphipod Crustacea (Talorchestia, &c.) which have abandoned a purely aquatic life. This view has been accepted and supported by Professors Korschelt and Heider (16). An important fact in its favour was discovered by Laurie (17), who investigated the embryology of two species of Scorpio under Lankester’s direction. It appears that the Malpighian tubes of Scorpio are developed from the mesenteron, viz. that portion of the gut which is formed by the hypoblast, whereas in Hexapod insects the similar caecal tubes are developed from the proctodaeum or in-pushed portion of the gut which is formed from epiblast. In fact it is not possible to maintain that the renal excretory tubes of the gut are of one common origin in the Arthropoda. They have appeared independently in connexion with a change in the excretion of nitrogenous waste in Arachnids, Crustacea, and the other classes of Arthropoda when aerial, as opposed to aquatic, respiration has been established—and they have been formed in some cases from the mesenteron, in other cases from the proctodaeum. Their appearance in the air-breathing Arachnids does not separate those forms from the water-breathing Arachnids which are devoid of them, any more than does their appearance in certain Amphipoda separate those Crustaceans from the other members of the class.
Further, it is pointed out by Korschelt and Heider that the hinder portion of the gut frequently acts in Arthropoda as an organ of nitrogenous excretion in the absence of any special excretory tubules, and that the production of such caeca from its surface in separate lines of descent does not involve any elaborate or unlikely process of growth. In other words, the Malpighian tubes of the terrestrial Arachnida are homoplastic with those of Hexapoda and Myriapoda, and not homogenetic with them. We are compelled to take a similar view of the agreement between the tracheal air-tubes of Arachnida and other tracheate Arthropods. They are homoplasts (see 18) one of another, and do not owe their existence in the various classes compared to a common inheritance of an ancestral tracheal system.
Conclusions arising from the Close Affinity of Limulus and Scorpio.—When we consider the relationships of the various classes of Arthropoda, having accepted and established the fact of the close genetic affinity of Limulus and Scorpio, we are led to important conclusions. In such a consideration we have to make use not only of the fact just mentioned, but of three important generalizations which serve as it were as implements for the proper estimation of the relationships of any series of organic forms. First of all there is the generalization that the relationships of the various forms of animals (or of plants) to one another is that of the ultimate twigs of a much-branching genealogical tree. Secondly, identity of structure in two organisms does not necessarily indicate that the identical structure has been inherited from an ancestor common to the two organisms compared (homogeny), but may be due to independent development of a like structure in two different lines of descent (homoplasy). Thirdly, those members of a group which, whilst exhibiting undoubted structural characters indicative of their proper assignment to that group, yet are simpler than and inferior in elaboration of their organization to other members of the group, are not necessarily representatives of the earlier and primitive phases in the development of the group—but are very often examples of retrogressive change or degeneration. The second and third implements of analysis above cited are of the nature of cautions or checks. Agreements are not necessarily due to common inheritance; simplicity is not necessarily primitive and ancestral.
On the other hand, we must not rashly set down agreements as due to “homoplasy” or “convergence of development” if we find two or three or more concurrent agreements. The probability is against agreement being due to homoplasy when the agreement involves a number of really separate (not correlated) coincidences. Whilst the chances are in favour of some one homoplastic coincidence or structural agreement occurring between some member or other of a large group a and some member or other of a large group b, the matter is very different when by such an initial coincidence the two members have been particularized. The chances against these two selected members exhibiting another really independent homoplastic agreement are enormous: let us say 10,000 to 1. The chances against yet another coincidence are a hundred million to one, and against yet one more “coincidence” they are the square of a hundred million to one. Homoplasy can only be assumed when the coincidence is of a simple nature, and is such as may be reasonably supposed to have arisen by the action of like selective conditions upon like material in two separate lines of descent.[4]
So, too, degeneration is not to be lightly assumed as the explanation of a simplicity of structure. There is a very definite criterion of the simplicity due to degeneration, which can in most cases be applied. Degenerative simplicity is never uniformly distributed over all the structures of the organism. It affects many or nearly all the structures of the body, but leaves some, it may be only one, at a high level of elaboration and complexity. Ancestral simplicity is more uniform, and does not co-exist with specialization and elaboration of a single organ. Further: degeneration cannot be inferred safely by the examination of an isolated case; usually we obtain a series of forms indicating the steps of a change in structure—and what we have to decide is whether the movement has been from the simple to the more complex, or from the more complex to the simple. The feathers of a peacock afford a convenient example of primitive and degenerative simplicity. The highest point of elaboration in colour, pattern and form is shown by the great eye-painted tail feathers. From these we can pass by gradual transitions in two directions, viz. either to the simple lateral tail feathers with a few rami only, developed only on one side of the shaft and of uniform metallic coloration—or to the simple contour feathers of small size, with the usual symmetrical series of numerous rami right and left of the shaft and no remarkable colouring. The one-sided specialization and the peculiar metallic colouring of the lateral tail feathers mark them as the extreme terms of a degenerative series, whilst the symmetry, likeness of constituent parts inter se, and absence of specialized pigment, as well as the fact that they differ little from any average feather of birds in general, mark the contour feather as primitively simple, and as the starting-point from which the highly elaborated eye-painted tail feather has gradually evolved.
Applying these principles to the consideration of the Arachnida, we arrive at the conclusion that the smaller and simpler Arachnids are not the more primitive, but that the Acari or mites are, in fact, a degenerate group. This was maintained by Lankester in 1878 (19), again in 1881 (20); it was subsequently announced as a novelty by Claus in 1885 (21). Though the aquatic members of a class of animals are in some instances derived from terrestrial forms, the usual transition is from an aquatic ancestry to more recent land-living forms. There is no doubt, from a consideration of the facts of structure, that the aquatic water-breathing Arachnids, represented in the past by the Eurypterines and to-day by the sole survivor Limulus, have preceded the terrestrial air-breathing forms of that group. Hence we see at once that the better-known Arachnida form a series, leading from Limulus-like aquatic creatures through scorpions, spiders and harvest-men, to the degenerate Acari or mites. The spiders are specialized and reduced in apparent complexity, as compared with the scorpions, but they cannot be regarded as degenerate since the concentration of structure which occurs in them results in greater efficiency and power than are exhibited by the scorpion. The determination of the relative degree of perfection of organization attained by two animals compared is difficult when we introduce, as seems inevitable, the question of efficiency and power, and do not confine the question to the perfection of morphological development. We have no measure of the degree of power manifested by various animals—though it would be possible to arrive at some conclusions as to how that “power” should be estimated. It is not possible here to discuss that matter further. We must be content to point out that it seems that the spiders, the pedipalps, and other large Arachnids have not been derived from the scorpions directly, but have independently developed from aquatic ancestors, and from one of these independent groups—probably through the harvest-men from the spiders—the Acari have finally resulted.
Leaving that question for consideration in connexion with the systematic statement of the characters of the various groups of Arachnida which follows on p. 299, it is well now to consider the following question, viz., seeing that Limulus and Scorpio are such highly developed and specialized forms, and that they seem to constitute as it were the first and second steps in the series of recognized Arachnida—what do we know, or what are we led to suppose with regard to the more primitive Arachnida from which the Eurypterines and Limulus and Scorpio have sprung? Do we know in the recent or fossil condition any such primitive Arachnids? Such a question is not only legitimate, but prompted by the analogy of at least one other great class of Arthropods. The great Arthropod class, the Crustacea, presents to the zoologist at the present day an immense range of forms, comprising the primitive phyllopods, the minute copepods, the parasitic cirrhipedes and the powerful crabs and lobsters, and the highly elaborated sand-hoppers and slaters. It has been insisted, by those who accepted Lankester’s original doctrine of the direct or genetic affinity of the Chaetopoda and Arthropoda, that Apus and Branchipus really come very near to the ancestral forms which connected those two great branches of Appendiculate (Parapodiate) animals. On the other hand, the land crabs are at an immense distance from these simple forms. The record of the Crustacean family-tree is, in fact, a fairly complete one—the lower primitive members of the group are still represented by living forms in great abundance. In the case of the Arachnida, if we have to start their genealogical history with Limulus and Scorpio, we are much in the same position as we should be in dealing with the Crustacea, were the whole of the Entomostraca and the whole of the Arthrostraca wiped out of existence and record. There is no possibility of doubt that the series of forms corresponding in the Arachnidan line of descent, to the forms distinguished in the Crustacean line of descent as the lower grade—the Entomostraca—have ceased to exist, and not only so, but have left little evidence in the form of fossils as to their former existence and nature. It must, however, be admitted as probable that we should find some evidence, in ancient rocks or in the deep sea, of the early more primitive Arachnids. And it must be remembered that such forms must be expected to exhibit, when found, differences from Limulus and Scorpio as great as those which separate Apus and Cancer. The existing Arachnida, like the higher Crustacea, are “nomomeristic,” that is to say, have a fixed typical number of somites to the body. Further, they are like the higher Crustacea, “somatotagmic,” that is to say, they have this limited set of somites grouped in three (or more) “tagmata” or regions of a fixed number of similarly modified somites—each tagma differing in the modification of its fixed number of somites from that characterizing a neighbouring “tagma.” The most primitive among the lower Crustacea, on the other hand, for example, the Phyllopoda, have not a fixed number of somites, some genera—even allied species—have more, some less, within wide limits; they are “anomomeristic.” They also, as is generally the case with anomomeristic animals, do not exhibit any conformity to a fixed plan of “tagmatism” or division of the somites of the body into regions sharply marked off from one another; the head or prosomatic tagma is followed by a trunk consisting of somites which either graduate in character as we pass along the series or exhibit a large variety in different genera, families and orders, of grouping of the somites. They are anomotagmic, as well as anomomeristic.
When it is admitted—as seems to be reasonable—that the primitive Arachnida would, like the primitive Crustacea, be anomomeristic and anomotagmic, we shall not demand of claimants for the rank of primitive Arachnids agreement with Limulus and Scorpio in respect of the exact number of their somites and the exact grouping of those somites; and when we see how diverse are the modifications of the branches of the appendages both in Arachnida and in other classes of Arthropoda (q.v.), we shall not over-estimate a difference in the form of this or that appendage exhibited by the claimant as compared with the higher Arachnids. With those considerations in mind, the claim of the extinct group of the trilobites to be considered as representatives of the lower and more primitive steps in the Arachnidan genealogy must, it seems, receive a favourable judgment. They differ from the Crustacea in that they have only a single pair of prae-oral appendages, the second pair being definitely developed as mandibles. This fact renders their association with the Crustacea impossible, if classification is to be the expression of genetic affinity inferred from structural coincidence. On the contrary, this particular point is one in which they agree with the higher Arachnida. But little is known of the structure of these extinct animals; we are therefore compelled to deal with such special points of resemblance and difference as their remains still exhibit. They had lateral eyes[5] which resemble no known eyes so closely as the lateral eyes of Limulus. The general form and structure of their prosomatic carapace are in many striking features identical with that of Limulus. The trilobation of the head and body—due to the expansion and flattening of the sides or “pleura” of the tegumentary skeleton—is so closely repeated in the young of Limulus that the latter has been called “the trilobite stage” of Limulus (fig. 42 compared with fig. 41). No Crustacean exhibits this trilobite form. But most important of the evidences presented by the trilobites of affinity with Limulus, and therefore with the Arachnida, is the tendency less marked in some, strongly carried out in others, to form a pygidial or telsonic shield—a fusion of the posterior somites of the body, which is precisely identical in character with the metasomatic carapace of Limulus. When to this is added the fact that a post-anal spine is developed to a large size in some trilobites (fig. 38), like that of Limulus and Scorpio, and that lateral spines on the pleura of the somites are frequent as in Limulus, and that neither metasomatic fusion of somites nor post-anal spine, nor lateral pleural spines are found in any Crustacean, nor all three together in any Arthropod besides the trilobites and Limulus—the claim of the trilobites to be considered as representing one order of a lower grade of Arachnida, comparable to the grade Entomostraca of the Crustacea, seems to be established.
The fact that the single pair of prae-oral appendages of trilobites, known only as yet in one genus, is in that particular case a pair of uni-ramose antennae—does not render the association of trilobites and Arachnids improbable. Although the prae-oral pair of appendages in the higher Arachnida is usually chelate, it is not always so; in spiders it is not so; nor in many Acari. The bi-ramose structure of the post-oral limbs, demonstrated by Beecher in the trilobite Triarthrus, is no more inconsistent with its claim to be a primitive Arachnid than is the foliaceous modification of the limbs in Phyllopods inconsistent with their relationship to the Arthrostracous Crustaceans such as Gammarus and Oniscus.
Thus, then, it seems that we have in the trilobites the representatives of the lower phases of the Arachnidan pedigree. The simple anomomeristic trilobite, with its equi-formal somites and equi-formal appendages, is one term of the series which ends in the even more simple but degenerate Acari. Between the two and at the highest point of the arc, so far as morphological differentiation is concerned, stands the scorpion; near to it in the trilobite’s direction (that is, on the ascending side) are Limulus and the Eurypterines—with a long gap, due to obliteration of the record, separating them from the trilobite. On the other side—tending downwards from the scorpion towards the Acari—are the Pedipalpi, the spiders, the book-scorpions, the harvest-men and the water-mites.
The strange nobody-crabs or Pycnogonids occupy a place on the ascending half of the arc below the Eurypterines and Limulus. They are strangely modified and degenerate, but seem to be (as explained in the systematic review) the remnant of an Arachnidan group holding the same relation to the scorpions which the Laemodipoda hold to the Podophthalmate Crustacea.
We have now to offer a classification of the Arachnida and to pass in review the larger groups, with a brief statement of their structural characteristics.
In the bibliography at the close of this article (referred to by leaded arabic numerals in brackets throughout these pages), the titles of works are given which contain detailed information as to the genera and species of each order or sub-order, their geographical distribution and their habits and economy so far as they have been ascertained. The limits of space do not permit of a fuller treatment of those matters here.
- Tabular Classification[6] of the Arachnida.
- Class. ARACHNIDA.
Grade A. ANOMOMERISTICA.
- Sub-Class. TRILOBITAE.
- Orders. Not satisfactorily determined.
- Sub-Class. TRILOBITAE.
Grade B. NOMOMERISTICA.
- Sub-Class I. PANTOPODA.
- Order 1. Nymphonomorpha.
- Order 2. Ascorhynchomorpha.
- Order 3. Pycnogonomorpha.
- Sub-Class II. EU-ARACHNIDA.
- Grade a. delobranchia, Lankester (vel hydropneustea, Pocock).
- Order 1. Xiphosura.
- Order 2. Gigantostraca.
- Grade b. embolobranchia, Lankester (vel aeropneustea, Pocock).
- Section α. Pectinifera.
- Grade b. embolobranchia, Lankester (vel aeropneustea, Pocock).
- Order 1. Scorpionidea.
- Sub-order a. Apoxypoda.
- Sub-order b. Dionychopoda.
- Section β. Epectinata.}
- Order 2. Pedipalpi.
- Sub-order a. Uropygi.
- Tribe 1. Urotricha.
- Tribe 2. Tartarides.
- Sub-order b. Amblypygi.
- Sub-order a. Uropygi.
- Order 3. Araneae.
- Sub-order a. Mesothelae.
- Sub-order b. Opisthothelae.
- Tribe 1. Mygalomorphae.
- Tribe 2. Arachnomorphae.
- Order 4. Palpigradi (= Microthelyphonidae).
- Order 5. Solifugae (= Mycetophorae).
- Order 6. Pseudoscorpiones (= Chelonethi).
- Sub-order a. Panctenodactyli.
- Sub-order b. Hemirtenodactyli.
- Order 7. Podogona (= Ricinulel).
- Order 8. Opiliones.
- Sub-order a. Laniatores.
- Sub-order b. Palpatores.
- Sub-order c. Anepignathi.
- Order 9. Rhynchostomi (= Acari).
- Sub-order a. Notostigmata.
- Sub-order b. Cryptostigmata.
- Sub-order c. Metastigmata.
- Sub-order d. Prostigmata.
- Sub-order e. Astigmata.
- Sub-order f. Vermiformia.
- Sub-order g. Tetrapoda.
- Sub-Class I. PANTOPODA.
Class. ARACHNIDA.—Euarthropoda having two prosthomeres (somites which have passed from a post-oral to a prae-oral position), the appendages of the first represented by eyes, of the second by solitary rami which are rarely antenniform, more usually chelate. A tendency is exhibited to the formation of a metasomatic as well as a prosomatic carapace by fusion of the tergal surfaces of the somites. Intermediate somites forming a mesosoma occur, but tend to fuse superficially with the metasomatic carapace or to become co-ordinated with the somites of the metasoma, whether fused or distinct to form one region, the opisthosoma (abdomen of authors). In the most highly developed forms the two anterior divisions (tagmata) of the body, prosoma and mesosoma, each exhibit six pairs of limbs, pediform and plate-like respectively, whilst the metasoma consists of six limbless somites and a post-anal spine. The genital apertures are placed in the first somite following the prosoma, excepting where a praegenital somite, usually suppressed, is retained. Little is known of the form of the appendages in the lowest archaic Arachnida, but the tendency of those of the prosomatic somites has been (as in the Crustacea) to pass from a generalized bi-ramose or multi-ramose form to that of uni-ramose antennae, chelae and walking legs.
The Arachnida are divisible into two grades of structure—according to the fixity or non-fixity of the number of somites building up the body:—
Grade A (of the Arachnida). ANOMOMERISTICA.—Extinct archaic Arachnida, in which (as in the Entomostracous Crustacea) the number of well-developed somites may be more or less than eighteen and may be grouped only as head (prosoma) and trunk or may be further differentiated. A telsonic tergal shield of greater or less size is always present, which may be imperfectly divided into well-marked but immovable tergites indicating incompletely differentiated somites. The single pair of palpiform appendages in front of the mouth has been found in one instance to be antenniform, whilst the numerous post-oral appendages in the same genus were bi-ramose. The position of the genital apertures is not known. Compound lateral eyes present; median eyes wanting. The body and head have the two pleural regions of each somite flattened and expanded on either side of the true gut-holding body-axis. Hence the name of the sub-class signifying tri-lobed, a condition realized also in the Xiphosurous Arachnids. The members of this group, whilst resembling the lower Crustacea (as all lower groups of a branching genealogical tree must do), differ from them essentially in that the head exhibits only one prosthomere (in addition to the eye-bearing prosthomere) with palpiform appendages (as in all Arachnida) instead of two. The Anomomeristic Arachnida form a single sub-class, of which only imperfect fossil remains are known.
Sub-class (of the Anomomeristica). TRILOBITAE.—The single sub-class Trilobitae constitutes the grade Anomomeristica. It has been variously divided into orders by a number of writers. The greater or less evolution and specialization of the metasomatic carapace appears to be the most important basis for classification—but this has not been made use of in the latest attempts at drawing up a system of the Trilobites. The form of the middle and lateral regions of the prosomatic shield has been used, and an excessive importance attached to the demarcation of certain areas in that structure. Sutures are stated to mark off some of these pieces, but in the proper sense of that term as applied to the skeletal structures of the Vertebrata, no sutures exist in the chitinous cuticle of Arthropoda. That any partial fusion of originally distinct chitinous plates takes place in the cephalic shield of Trilobites, comparable to the partial fusion of bony pieces by suture in Vertebrata, is a suggestion contrary to fact.
The Trilobites are known only as fossils, mostly Silurian and prae-Silurian; a few are found in Carboniferous and Permian strata. As many as two thousand species are known. Genera with small metasomatic carapace, consisting of three to six fused segments distinctly marked though not separated by soft membrane, are Harpes, Paradoxides and Triarthrus (fig. 34). In Calymene, Homalonotus and Phacops (fig. 38) from six to sixteen segments are clearly marked by ridges and grooves in the metasomatic tagma, whilst in Illaenus the shield so formed is large but no somites are marked out on its surface. In this genus ten free somites (mesosoma) occur between the prosomatic and metasomatic carapaces. Asaphus and Megalaspis (fig. 39) are similarly constituted. In Agnostus (fig. 40) the anterior and posterior carapaces constitute almost the entire body, the two carapaces being connected by a mid-region of only two free somites. It has been held that the forms with a small number of somites marked in the posterior carapace and numerous free somites between the anterior and posterior carapace, must be considered as anterior to those in which a great number of posterior somites are traceable in the metasomatic carapace, and that those in which the traces of distinct somites in the posterior or metasomatic carapace are most completely absent must be regarded as derived from those in which somites are well marked in the posterior carapace and similar in appearance to the free somites. The genus Agnostus, which belongs to the last category, occurs abundantly in Cambrian strata and is one of the earliest forms known. This would lead to the supposition that the great development of metasomatic carapace is a primitive and not a late character, were it not for the fact that Paradoxides and Atops, with an inconspicuous telsonic carapace and numerous free somites, are also Cambrian in age, the latter indeed anterior in horizon to Agnostus.
On the other hand, it may well be doubted whether the pygidial or posterior carapace is primarily due to a fusion of the tergites of somites which were previously movable and well developed. The posterior carapace of the Trilobites and of Limulus is probably enough in origin a telsonic carapace—that is to say, is the tergum of the last segment of the body which carries the anus. From the front of this region new segments are produced in the first instance, and are added during growth to the existing series. This telson may enlarge, it may possibly even become internally and sternally developed as partially separate somites, and the tergum may remain without trace of somite formation, or, as appears to be the case in Limulus, the telson gives rise to a few well-marked somites (mesosoma and two others) and then enlarges without further trace of segmentation, whilst the chitinous integument which develops in increasing thickness on the terga as growth advances welds together the unsegmented telson and the somites in front of it, which were previously marked by separate tergal thickenings. It must always be remembered that we are liable (especially in the case of fossilized integuments) to attach an unwarranted interpretation to the mere discontinuity or continuity of the thickened plates of chitinous cuticle on the back of an Arthropod. These plates may fuse, and yet the somites to which they belong may remain distinct, and each have its pair of appendages well developed. On the other hand, an unusually large tergal plate, whether terminal or in the series, is not always due to fusion of the dorsal plates of once-separate somites, but is often a case of growth and enlargement of a single somite without formation of any trace of a new somite. For the literature of Trilobites see (22*).
Grade B (of the Arachnida) NOMOMERISTICA.—Arachnida in which, excluding from consideration the eye-bearing prosthomere, the somites are primarily (that is to say, in the common ancestor of the grade) grouped in three regions of six—(a) the “prosoma” with palpiform appendages, (b) the “mesosoma” with plate-like appendages, and (c) the “metasoma” with suppressed appendages. A somite placed between the prosoma and mesosoma—the prae-genital somite—appears to have belonged originally to the prosomatic series (which with its ocular prosthomere and palpiform limbs [Pantopoda], would thus consist of eight somites), but to have been gradually reduced. In living Arachnids, excepting the Pantopoda, it is either fused (with loss of its appendages) with the prosoma (Limulus,[7] Scorpio), after embryonic appearance, or is retained as a rudimentary, separate, detached somite in front of the mesosoma, or disappears altogether (excalation). The atrophy and total disappearance of ancestrally well-marked somites frequently take place (as in all Arthropoda) at the posterior extremity of the body, whilst excalation of somites may occur at the constricted areas which often separate adjacent “regions,” though there are very few instances in which it has been recognized. Concentration of the organ-systems by fusion of neighbouring regions (prosoma, mesosoma, metasoma), previously distinct, has frequently occurred, together with obliteration of the muscular and chitinous structures indicative of distinct somites. This concentration and obliteration of somites, often accompanied by dislocation of important segmental structures (such as appendages and nerve-ganglia), may lead to highly developed specialization (individuation, H. Spencer), as in the Araneae and Opiliones, and, on the other hand, may terminate in simplification and degeneration, as in the Acari.}
 |
Fig. 42.—So-called “trilobite stage” of Limulus polyphemus. A, Dorsal; B, ventral view. (From Korschelt and Heider, after Leuckart.) |
The most important general change which has affected the structure of the nomomeristic Arachnida in the course of their historic development is the transition from an aquatic to a terrestrial life. This has been accompanied by the conversion of the lamelliform gill-plates into lamelliform lung-plates, and later the development from the lung-chambers, and at independent sites, of tracheae or air-tubes (by adaptation of the vasifactive tissue of the blood-vessels) similar to those independently developed in Peripatus, Diplopoda, Hexapoda and Chilopoda. Probably tracheae have developed independently by the same process in several groups of tracheate Arachnids. The nomomeristic Arachnids comprise two sub-classes—one a very small degenerate offshoot from early ancestors; the other, the great bulk of the class.
Sub-Class I. (of the Nomomeristica). PANTOPODA.—Nomomeristic Arachnids, in which the somites corresponding to mesosoma and metasoma have entirely aborted. The seventh, and sometimes the eighth, leg-bearing somite is present and has its leg-like appendages fully developed. Monomeniscous eyes with a double (really triple) cell-layer formed by invagination, as in the Eu-arachnida, are present The Pantopoda stand in the same relation to Limulus and Scorpio that Cyamus holds to the thoracostracous Crustacea. The reduction of the organism to seven leg-bearing somites, of which the first pair, as in so many Eu-arachnida, are chelate, is a form of degeneration connected with a peculiar quasi-parasitic habit resembling that of the crustacean Laemodipoda. The genital pores are situate at the base of the 7th pair of limbs, and may be repeated on the 4th, 5th, and 6th. In all known Pantopoda the size of the body is quite minute as compared with that of the limbs: the alimentary canal sends a long caecum into each leg (cf. the Araneae) and the genital products are developed in gonocoels also placed in the legs.
The Pantopoda are divided into three orders, the characters of which are dependent on variation in the presence of the full number of legs.
Order 1. (of the Pantopoda). Nymphonomorpha, Pocock (nov.) (fig. 43).—In primitive forms belonging to the family Nymphonidae the full complement of appendages is retained—the 1st (mandibular), the 2nd (palpiform), and the 3rd (ovigerous) pairs being well developed in both sexes. In certain derivative forms constituting the family Pallenidae, however, the appendages of the 2nd pair are either rudimentary or atrophied altogether.
Two families: 1. Nymphonidae (genus Nymphon), and 2. Pallenidae (genus Pallene).
Order 2. Ascorhynchomorpha, Pocock (nov.).—Appendages of the 2nd and 3rd pairs retained and developed, as in the more primitive types of Nymphonomorpha; but those of the 1st pair are either rudimentary, as in the Ascorhynchidae, or atrophied, as in the Colossendeidae. In the latter a further specialization is shown in the fusion of the body segments.
Two families. 1. Ascorhynchidae (genera Ascorhynchus and Ammothea); 2. Colossendeidae (genera Colossendeis and Discoarachne).
Order 3. Pycnogonomorpha, Pocock (nov.).—Derivative forms in which the reduction in number of the anterior appendages is carried farther than in the other orders, reaching its extreme in the Pycnogonidae, where the 1st and 2nd pairs are absent in both sexes, and the 3rd pair also are absent in the female. In the Hannoniidae, however, which resemble the Pycnogonidae in the absence of the 3rd pair in the female and of the 2nd pair in both sexes, the 1st pair are retained in both sexes.
Two families: 1. Hannoniidae (genus Hannonia); 2. Pycnogonidae (genera Pycnogonum and Phoxichilus).
Remarks.—The Pantopoda are not known in the fossil condition. They are entirely marine, and are not uncommon in the coralline zone of the sea-coast. The species are few, not more than fifty (23). Some large species of peculiar genera are taken at great depths. Their movements are extremely sluggish. They are especially remarkable for the small size of the body and the extension of viscera into the legs. Their structure is eminently that of degenerate forms. Many frequent growths of coralline Algae and hydroid polyps, upon the juices of which they feed, and in some cases a species of gall is produced in hydroids by the penetration of the larval Pantopod into the tissues of the polyp.
Sub-Class II. (of the Nomomeristic Arachnida). EU-ARACHNIDA.—These start from highly developed and specialized aquatic branchiferous forms, exhibiting a prosoma with six pediform pairs of appendages, an intermediate prae-genital somite, a mesosoma of six somites bearing lamelliform pairs of appendages, and a metasoma of six somites devoid of appendages, and the last provided with a post-anal spine. Median eyes are present, which are monomeniscous, with distinct retinal and corneagenous cell-layers, and placed centrally on the prosoma. Lateral eyes also may be present, arranged in lateral groups, and having a single or double cell-layer beneath the lens. The first pair of limbs is often chelate or prehensile, rarely antenniform; whilst the second, third and fourth may also be chelate, or may be simple palps or walking legs.
An internal skeletal plate, the so-called “entosternite” of fibro-cartilaginous tissue, to which many muscles are attached, is placed between the nerve-cords and the alimentary tract in the prosoma of the larger forms (Limulus, Scorpio, Mygale). In the same and other leading forms a pair of much-coiled glandular tubes, the coxal glands (coelomocoels in origin), is found with a duct opening on the coxa of the fifth pair of appendages of the prosoma. The vascular system is highly developed (in the non-degenerate forms); large arterial branches closely accompany or envelop the chief nerves; capillaries are well developed. The blood-corpuscles are large amoebiform cells, and the blood-plasma is coloured blue by haemocyanin. The alimentary canal is uncoiled and cylindrical, and gives rise laterally to large gastric glands, which are more than a single pair in number (two to six pairs), and may assume the form of simple caeca. The mouth is minute and the pharynx is always suctorial, never gizzard-like. The gonadial tubes (gonocoels or gonadial coelom) are originally reticular and paired, though they may be reduced to a simpler condition. They open on the first somite of the mesosoma. In the numerous degenerate forms simplification occurs by obliteration of the demarcations of somites and the fusion of body-regions, together with a gradual suppression of the lamelliferous respiratory organs and the substitution for them of tracheae, which, in their turn, in the smaller and most reduced members of the group, may also disappear. The Eu-arachnida are divided into two grades with reference to the condition of the respiratory organs as adapted to aquatic or terrestrial life.
Grade a (of the Eu-arachnida). delobranchia (Hydropheustea).
Mesosomatic segments furnished with large plate-like appendages, the 1st pair acting as the genital operculum, the remaining pairs being provided with branchial lamellae fitted for breathing oxygen dissolved in water. The prae-genital somite partially or wholly obliterated in the adult. The mouth lying far back, so that the basal segments of all the prosomatic appendages, excepting those of the 1st pair, are capable of acting as masticatory organs. Lateral eyes consisting of a densely packed group of eye-units (“compound” eyes).
Order 1. Xiphosura.—The prae-genital somite fuses in the embryo with the prosoma and disappears (see fig. 19). Not free-swimming, none of the prosomatic appendages modified to act as paddles; segments of the mesosoma and metasoma ( = opisthosoma) not more than ten in number, distinct or coalesced.
- Family—Limulidae (Limulus).
- ” *Belinuridae (Belinurus, Aglaspis, Prestwichia).
- ” *Hemiaspidae (Hemiaspis, Bunodes).
 |
Fig. 44.—Dorsal view of Limulus polyphemus, Latr. |
Remarks.—The Xiphosura are marine in habit, frequenting the shore. They are represented at the present day by the single genus Limulus (figs. 44 and 45; also figs. 7, 9, 11, to 15 and 20), often termed the king-crab, which occurs on the American coast of the Atlantic Ocean, but not on its eastern coasts, and on the Asiatic coast of the Pacific. The Atlantic species (L. polyphemus) is common on the coasts of the United States, and is known as the king-crab or horse-shoe crab. A single specimen was found in the harbour of Copenhagen in the 18th century, having presumably been carried over by a ship to which it clung.
A species of Limulus is found in the Buntersandstein of the Vosges; L. Walchi is abundant in the Oolitic lithographic slates of Bavaria.
The genera Belinurus, Aglaspis, Prestwichia, Hemiaspis and Bunodes consist of small forms which occur in Palaeozoic rocks. In none of them are the appendages known, but in the form of the two carapaces and the presence of free somites they are distinctly intermediate between Limulus and the Trilobitae. The young form of Limulus itself (fig. 40) is also similar to a Trilobite so far as its segmentation and trilobation are concerned. The lateral eyes of Limulus appear to be identical in structure and position with those of certain Trilobitae.
Order 2. Gigantostraca (figs. 46, 47).—Free-swimming forms, with the appendages of the 6th or 5th and 6th pairs flattened or lengthened to act as oars; segments of mesosoma and metasoma (= opisthosoma), twelve in number.
Appendages of anterior pair very large and chelate.
- Sub-order Pterygotomorpha, Pterygotidae (Pterygotus).
Appendages of anterior pair minute and chelate.
| Sub-order Eurypteromorpha | Stylonuridae (Stylonurus). Eurypteridae (Eurypterus, Slimonia). |
Remarks.—The Gigantostraca are frequently spoken of as “the Eurypterines.” Not more than thirty species are known. They became extinct in Palaeozoic times, and are chiefly found in the Upper Silurian, though extending upwards as far as the Carboniferous. They may be regarded as “macrourous” Xiphosura; that is to say, Xiphosura in which the nomomeristic number of eighteen well-developed somites is present and the posterior ones form a long tail-like region of the body. There still appears to be some doubt whether in the sub-order Eurypteromorpha the first pair of prosomatic appendages (fig. 46) is atrophied, or whether, if present, it has the form of a pair of tactile palps or of minute chelae. Though there are indications of lamelliform respiratory appendages on mesosomatic somites following that bearing the genital operculum, we cannot be said to have any proper knowledge as to such appendages, and further evidence with regard to them is much to be desired. (For literature see Zittel, 22*.)
Grade b (of the Eu-arachnida). embolobranchia (Aeropneustea).
 |
Restored after Thorell’s indications by R. I. Pocock. Fig. 48.—Dorsal view of a restoration of Palaeophonus nuncius, Thorell. The Silurian scorpion from Gothland. |
In primitive forms the respiratory lamellae of the appendages of the 3rd, 4th, 5th and eth, or of the 1st and 2nd mesosomatic somites are sunk beneath the surface of the body, and become adapted to breathe atmospheric oxygen, forming the leaves of the so-called lung-books. In specialized forms these pulmonary sacs are wholly or partly replaced by tracheal tubes. The appendages of the mesosoma generally suppressed; in the more primitive forms one or two pairs may be retained as organs subservient to reproduction or silk-spinning. Mouth situated more forwards than in Delobranchia, no share in mastication being taken by the basal segments of the 5th and 6th pairs of prosomatic appendages. Lateral eyes, when present, represented by separate ocelli.
The prae-genital somite, after appearing in the embryo, either is obliterated (Scorpio, Galeodes, Opilio and others) or is retained as a reduced narrow region of the body, the “waist,” between prosoma and mesosoma. It is represented by a full-sized tergal plate in the Pseudo-scorpiones.
Section α. Pectinifera.—The primitive distinction between the mesosoma and the metasoma retained, the latter consisting of six somites and the former of six somites in the adult, each of which is furnished during growth with a pair of appendages. Including the prae-genital somite (fig. 16), which is suppressed in the adult, there are thirteen somites behind the prosoma. The appendages of the 1st and 2nd mesosomatic somites persisting as the genital operculum and pectones respectively, those of the 3rd, 4th, 5th and 6th somites (? in Palaeophonus) sinking below the surface during growth in connexion with the formation of the four pairs of pulmonary sacs (see fig. 17). Lateral eyes monostichous.
Order 1. Scorpiones.—Prosoma covered by a single dorsal shield, bearing typically median and lateral eyes; its sternal elements reduced to a single plate lodged between or behind the basal segments of the 5th and 6th pairs of appendages. Appendages of 1st pair tri-segmented, chelate; of 2nd pair chelate, with their basal segments subserving mastication; of 3rd, 4th, 5th and 6th pairs similar in form and function, except that in recent and Carboniferous forms the basal segments of the 3rd and 4th are provided with sterno-coxal (maxillary) lobes, those of the 4th pair meeting in the middle line and underlying the mouth. The five posterior somites of the metasoma constricted to form a “tail,” the post-anal sclerite persisting as a weapon of offence and provided with a pair of poison glands (see figs. 8, 10, 12, 13, 14, 15, 21 and 22).
Sub-order Apoxypoda.—The 3rd, 4th, 5th and 6th pairs of appendages short, stout, tapering, the segments about as wide as long, except the apical, which is distally slender, pointed, slightly curved, and without distinct movable claws.
Family—Palaeophonidae, Palaeophonus (figs. 48 and 49).
Sub-order Dionychopoda.—The 3rd, 4th, 5th and 6th pairs of appendages slender, not evenly tapering, the segments longer than wide; the apical segment short, distally truncate, and provided with a pair of movable claws. Basal segments of the 5th and 6th pairs of appendages abutting against the sternum of the prosoma (see fig. 10 and figs. 51, 52 and 53).
Family—Pandinidae (Pandinus, Opisthophthalmus, Urodacus).
” Vejovidae (Vaejovis, Jurus, Euscorpius, Broteas).
” Bothriuridae (Bothriurus, Cercophonius).
” Buthidae (Buthus, Centrurus).
| ” *Cyclophthalmidae (Cyclophthalmus) ” *Eoscorpiidae (Eoscorpius, Centromachus) |
Carboniferous. |
Remarks on the Order Scorpiones.—The Scorpion is one of the great animals of ancient lore and tradition. It and the crab are the only two invertebrates which had impressed the minds of early men sufficiently to be raised to the dignity of astronomical representation. It is all the more remarkable that the scorpion proves to be the oldest animal form of high elaboration which has persisted to the present day. In the Upper Silurian two specimens of a scorpion have been found (figs. 48, 49), one in Gothland and one in Scotland, which would be recognized at once as true scorpions by a child or a savage. The Silurian scorpion Palaeophonus, differs, so far as obvious points are concerned, from a modern scorpion only in the thickness of its legs and in their terminating in strong spike-like joints, instead of being slight and provided with a pair of terminal claws. The legs of the modern scorpion (fig. 10; fig. 51) are those of a terrestrial Arthropod, such as a beetle; whilst those of the Silurian scorpion are the legs of an aquatic Arthropod, such as a crab or lobster. It is probable that the Silurian scorpion was an aquatic animal, and that its respiratory lamellae were still projecting from the surface of the body to serve as branchiae. No trace of “stigmata,” the orifices of the lung-chambers of modern scorpions, can be found in the Scottish specimen of Palaeophonus, which presents the ventral surface of the animal to view. On the other hand, no trace of respiratory appendages excepting the pectens can be detected in the specimen (see fig. 49).

|
| From Lankester, Journ. Linn. Soc. Zool. vol. xvi., 1881. |
|
Fig. 51.—Drawing from life of the desert scorpion, Buthusaustralis, Lin., from Biskra, N. Africa. |
Fossil scorpions of the modern type are found in the Coal Measures. At the present day scorpions of various genera are found in all the warm regions of the world. In Europe they occur as far north as Bavaria and the south of France. The largest species measure 9 in. from the front of the head to the end of the sting, and occur in tropical India and Africa. Between 200 and 300 species are known. The scorpions use their large chelae for seizing prey and for fighting with one another. They never use the sting when (as frequently happens) they attack another scorpion, because, as was ascertained by A. G. Bourne (24), the poison exuded by the sting has no injurious effect on another scorpion nor on the scorpion itself. The stories of a scorpion stinging itself to death when placed in a circle of burning coals are due to erroneous observation. When placed in such a position the scorpion faints and becomes inert. It is found (Bourne, 24) that some species of scorpion faint at a temperature of 40° Cent. They recover on being removed to cooler conditions. A scorpion having seized its prey (usually a large insect, or small reptile or mammal) with the large chelae brings its tail over its head, and deliberately punctures the struggling victim twice with its sting (fig. 52). The poison of the sting is similar to snake-poison (Calmette), and rapidly paralyses animals which are not immune to it. It is probably only sickly adults or young children of the human race who can be actually killed by a scorpion’s sting. When the scorpion has paralysed its prey in this way, the two short chelicerae are brought into play (fig. 53). By the crushing action of their pincers, and an alternate backward and forward movement, they bring the soft blood-holding tissues of the victim close to the minute pin-hole aperture which is the scorpion’s mouth. The muscles acting on the bulb-like pharynx now set up a pumping action (see Huxley, 26); and the juices—but no solid matter, excepting such as is reduced to powder—are sucked into the scorpion’s alimentary canal. A scorpion appears to prefer for its food another scorpion, and will suck out the juices of an individual as large as itself. When this has taken place, the gorged scorpion becomes distended and tense in the mesosomatic region. It is certain that the absorbed juices do not occupy the alimentary canal alone, but pass also into its caecal off-sets which are the ducts of the gastric glands (see fig. 33).
All Arachnida, including Limulus, feed by suctorial action in essentially the same way as Scorpio.
Scorpions of various species have been observed to make a hissing noise when disturbed, or even when not disturbed. The sound is produced by stridulating organs developed on the basal joints of the limbs, which differ in position and character in different genera (see Pocock, 27). Scorpions copulate with the ventral surfaces in contact. The eggs are fertilized, practically in the ovary, and develop in situ. The young are born fully formed and are carried by the mother on her back. As many as thirty have been counted in a brood. For information as to the embryology of scorpions, the reader is referred to the works named in the bibliography below. Scorpions do not possess spinning organs nor form either snares or nests, so far as is known. But some species inhabiting sandy deserts form extensive burrows. The fifth pair of prosomatic appendages is used by these scorpions when burrowing, to kick back the sand as the burrow is excavated by the great chelae.
References to works dealing with the taxonomy and geographical distribution of scorpions are given at the end of this article (28).
Section β. Epectinata.—The primitive distinction between the mesosoma and the metasoma wholly or almost wholly obliterated, the two regions uniting to form an opisthosoma, which never consists of more than twelve somites and never bears appendages or breathing-organs behind the 4th somite. The breathing-organs of the opisthosoma, when present, represented by two pairs of stigmata, opening either upon the 1st and 2nd (Pedipalpi) or the 2nd and 3rd somites (Solifugae, Pseudo-scorpiones), or by a single pair upon the 3rd (? 2nd) somite (Opiliones) of the opisthosoma, there being rarely an additional stigma on the 4th (some Solifugae). The appendages of the 2nd somite of the opisthosoma absent, rarely minute and bud-like (some Amblypygi), never pectiniform. A prae-genital somite is often present either in a reduced condition forming a waist (Pedipalpi, Araneae, Palpigradi) or as a full-sized tergal plate (Pseudo-scorpiones); in some it is entirely atrophied (Solifugae, Holosomata, and Rhynchostomi). Lateral eyes when present diplostichous.
Remarks.—The Epectinate Arachnids do not stand so close to the aquatic ancestors of the Embolobranchia as do the Pectiniferous scorpions. At the same time we are not justified in supposing that the scorpions stand in any way as an intermediate grade between any of the existing Epectinata and the Delobranchia. It is probable that the Pedipalpi, Araneae, and Podogona have been separately evolved as distinct lines of descent from the ancient aquatic Arachnida. The Holosomata and Rhynchostomi are probably offshoots from the stem of the Araneae, and it is not unlikely (in view of the structure of the prosomatic somites of the Tartarides) that the Solifugae are connected in origin with the Pedipalpi. The appearance of tracheae in place of lung-sacs cannot be regarded as a starting-point for a new line of descent comprising all the tracheate forms; tracheae seem to have developed independently in different lines of descent. On the whole, the Epectinata are highly specialized and degenerate forms, though there are few, if any, animals which surpass the spiders in rapidity of movement, deadliness of attack and constructive instincts.
Order 2. Pedipalpi (figs. 54 to 59).—Appendages of 1st pair bisegmented, without poison gland; of 2nd pair prehensile, their basal segments underlying the proboscis, and furnished with sterno-coxal (maxillary) process, the apical segment tipped with a single movable or immovable claw; appendages of 3rd pair different from the remainder, tactile in function, with at least the apical segment many-jointed and clawless. The ventral surface of the prosoma bears prosternal, metasternal and usually mesosternal chitine-plates (fig. 55). A narrow prae-genital somite is present between opisthosoma and prosoma (figs. 55, 57). Opisthosoma consisting of eleven somites, almost wholly without visible appendages. Intromittent organ of male beneath the genital operculum (= sternum of the 1st somite of opisthosoma).
Note.—The possibility of another interpretation of the anterior somites of the mesosoma and the prae-genital somite must be borne in mind. Possibly, though not probably, the somites carrying the two lung-sacs correspond to the first two lung-bearing somites of Scorpio, and it is the genital opening which has shifted. The same caution applies in the case of the Araneae. Excalation of one or of two anterior mesosomatic somites, besides the prae-genital somite, would then have to be supposed to have occurred also.
Sub-order a. Uropygi.—Prosoma longer than wide, its sternal area very narrow, furnished with a large prosternal and metasternal plate, and often with a small mesosternal sclerite. Appendages of 2nd pair with their basal segments united in the middle line and incapable of lateral movement; appendages of 3rd pair with only the apical segment many-jointed. Opisthosoma without trace of appendages; its posterior somites narrowed to form a movable tail for the support of the post-anal sclerite, which has no poison glands.
Tribe 1. Urotricha.—Dorsal area of prosoma covered with a single shield (? two in Geralinura), bearing median and lateral eyes. Post-anal sclerite modified as a long, many-jointed feeler. Appendages of 2nd pair folding in a horizontal plane, completely chelate, the claw immovably united to the sixth segment. Respiratory organs present in the form of pulmonary sacs.
Family—Thelyphonidae (Thelyphonus (fig. 54), Hypoctonus, *Geralinura).
Tribe 2. Tartarides.—Small degenerate forms with the dorsal area of the prosoma furnished with two shields, a larger in front covering the anterior four somites, and a smaller behind covering the 5th and 6th somites; the latter generally subdivided into a right and left portion. There is also a pair of narrow tergal sclerites interposed between the anterior and posterior shields. Eyes evanescent or absent. Appendages of 2nd pair folding in a vertical plane, not chelate, the claw long and movable. Post-anal sclerite short and undivided. No distinct respiratory stigmata behind the sterna of the 1st and 2nd somites of the opisthosoma.
Family—Hubbardiidae (Schizomus, Hubbardia) (figs. 57-59).
Sub-order b. Amblypygi.—Prosoma wider than long, covered above by a single shield bearing median and lateral eyes, which have diplostichous ommatea. Sternal area broad, with prosternal, two mesosternal, and metasternal plates, the prosternum projecting forwards beneath the coxae of the 2nd pair of appendages. Appendages of 2nd pair folding in a horizontal plane; their basal segments freely movable; claw free or fused; basal segments of 4th and 5th pairs widely separated by the sternal area; appendages of 3rd pair with all the segments except the proximal three, forming a many-jointed flagellum. Opisthosoma without post-anal sclerite and posterior caudal elongation: with frequently a pair of small lobate appendages on the sternum of the 3rd somite. Respiratory organs, as in Urotricha.
- Family—Phrynichidae (Phrynichus, Damon).
- ” Admetidae (Admetus, Heterophrynus).
- ” Charontidae (Charon, Sarax).
- (Family ?)—*Graeophonus.
Remarks.—The Pedipalpi are confined to the tropics and warmer temperate regions of both hemispheres. Fossil forms occur in the Carboniferous. The small forms known as Schizomus and Hubbardia are of special interest from a morphological point of view. The Pedipalpi have no poison glands. (Reference to literature (29).)
Order 3. Araneae (figs. 60 to 64.).—Prosoma covered with a single shield and typically furnished with median and lateral eyes of diplostichous structure, as in the Amblypygi. The sternal surface wide, continuously chitinized, but with prosternal and metasternal elements generally distinguishable at the anterior and posterior ends respectively of the large mesosternurm. Prosternum underlying the proboscis. Appendages of 1st pair have two segments, as in Pedipalpi, but are furnished with poison gland, and are retroverts. Appendages of 2nd pair not underlying the mouth, but freely movable and, except in primitive forms, furnished with a maxillary lobe; the rest of the limb like the legs, tipped with a single claw and quite unmodified (except in ♂). Remaining pairs of appendages similar in form and function, each tipped with two or three claws. Opisthosoma when segmented showing the same number of somites as in the Pedipalpi; usually unsegmented, the prae-genital somite constricted to form the waist; the appendages of its 3rd and 4th somites retained as spinning mammillae. Respiratory organs (see fig. 63, stg), as in the Amblypygi, or with the posterior pair, rarely the anterior pair as well, replaced by tracheal tubes. Intromittent organ of male in the apical segment of the 2nd prosomatic appendage.
Sub-order a. Mesothelae (see figs. 60 to 62).—Opisthosoma distinctly segmented, furnished with 11 tergal plates, as in the Amblypygi; the ventral surface of the 1st and 2nd somites with large sternal plates, covering the genital aperture and the two pairs of pulmonary sacs, the sternal plates from the 6th to the 11th somites represented by integumental ridges, weakly chitinized in the middle. The two pairs of spinning appendages retain their primitive position in the middle of the lower surface of the opisthosoma far in advance of the anus on the 3rd and 4th somites, each appendage consisting of a stout, many-jointed outer branch and a slender, unsegmented inner branch. Prosoma as in the Mygalomorphae, except that the mesosternal area is long and narrow.
- Family—Liphistiidae (Liphistius, *Arthrolycosa).
Sub-order b. Opisthothelae (see fig. 63).—Opisthosoma without trace of separate terga and sterna, the segmentation merely represented posteriorly by slight integumental folds and the sterna of the 1st and 2nd somites by the opercular plates of the pulmonary sacs. The spinning appendages migrate to the posterior end of the opisthosoma and take up a position close to the anus; the inner branches of the anterior pair either atrophy or are represented homogenetically by a plate, the cribellum, or by an undivided membranous lobe, the colulus.
Tribe 1. Mygalomorphae.—The plane of the articulation of the appendages of the 1st pair to the prosoma (the retrovert) vertical, the basal segment projecting straight forwards at its proximal end, the distal segment or fang closing backwards in a direction subparallel to the long axis of the body. Two pairs of pulmonary sacs.
Families—Theraphosidae (Avicularia, Poecilotheria). Barychelidae (Barychelus, Plagiobothrus). Dipluridae (Diplura, Macrothele). Ctenizidae (Cteniza, Nemesia). Atypidae (Atypus, Calommata). Tribe 2. Arachnomorphae.—The plane of the articulation of the appendages of the 1st pair to the prosoma horizontal, the basal segment projecting vertically downwards, at least at its proximal end, the distal segment or fang closing inwards nearly or quite at right angles to the long axis of the body. The posterior pulmonary sacs (except in Hypochilus) replaced by tracheal tubes; the anterior and posterior pairs replaced by tracheal tubes in the Caponiidae. Principal families—Hypochilidae (Hypochilus). Dysderidae (Dysdera, Segestria). Caponiidae (Caponia, Nops). Filistatidae (Filistata). Uloboridae (Uloborus, Dinopis). Argiapidae (Nephila, Gasteracantha). Pholcidae (Pholcus, Artema). Agelenidae (Tegenaria). Lycosidae (Lycosa). Clubionidae (Clubiona, Olios, Sparassus) Gnaphosidae (Gnaphosa, Hemiclaea). Thomisidae (Thomisus). Attidae (Salticus). Urocteidae (Uroctea). Eresidae (Eresus).
Remarks on the Araneae.—The Spiders are the most numerous and diversified group of the Arachnida; about 2000 species are known. No noteworthy fossil spiders are known; the best-preserved are in amber of Oligocene age. Protolycosa and Arthrolycosa occur in the Carboniferous. Morphologically, the spiders are remarkable for the concentration and specialization of their structure, which is accompanied with high physiological efficiency. The larger species of Bird’s Nest Spiders (Avicularia), the opisthosoma of which is as large as a bantam’s egg, undoubtedly attack young birds, and M‘Cook gives an account of the capture in its web by an ordinary house spider of a small mouse. The “retrovert” or bent-back first pair of appendages is provided with a poison gland opening on the fang or terminal segment. Spiders form at least two kinds of constructions—snares for the capture of prey and nests for the preservation of the young. The latter are only formed by the female, which is a larger and more powerful animal than the male. Like the scorpions the spiders have a special tendency to cannibalism, and accordingly the male, in approaching the female for the purpose of fertilizing her, is liable to be fallen upon and sucked dry by the object of his attentions. The sperm is removed by the male from the genital aperture into a special receptacle on the terminal segment of the 2nd prosomatic appendage. Thus held out at some distance from the body, it is cautiously advanced by the male spider to the genital aperture of the female.
For an account of the courtship and dancing of spiders, of their webs and floating lines, the reader is referred to the works of M‘Cook (30) and the Peckhams (31), whilst an excellent account of the nests of trap-door spiders is given by Moggridge (32). References to systematic works will also be found at the end of this article (33).
Order 4. Palpigradi = Microthelyphonidae (see fig. 65).—Prosoma covered above by three plates, a larger representing the dorsal elements of the first four somites, and two smaller representing the dorsal elements of the 5th and 6th.
Its ventral surface provided with one prosternal, two mesosternal and one metasternal plate. Appendages of 1st pair consisting of three segments, completely chelate, without poison gland; of 2nd pair slender, leg-like, tipped with three claws, the basal segment without sterno-coxal process taking no share in mastication, and widely separated from its fellow of the opposite side; 3rd, 4th, 5th and 6th appendages similar in form to the 2nd and to each other.
Proboscis free, not supported from below by either the prosternum or the basal segments of the appendages of the 2nd pair.
(Original drawing by Pocock and Pickard-Cambridge, after Hansen and Sörensen.)
Opisthosoma consisting of only ten somites, which have no tergal and sternal elements, the prae-genital somite contracted to form a “waist,” as in the Pedipalpi; the last three narrowed to form a caudal support for the many-jointed flagelliform telson, as in the Urotricha. Respiratory organs atrophied.
Family—Koeneniidae (Koenenia).
Remarks.—An extremely remarkable minute form originally described by Grassi (34) from Sicily, and since further described by Hansen (35). Recently the genus has been found in Texas, U.S.A. Only one genus of the order is known.
Order 5. Solifugae = Mycetophorae (see figs. 66 to 69).—Dorsal area of prosoma covered with three distinct plates, two smaller representing the terga of the 5th and 6th somites, and a larger representing those of the anterior four somites, although the reduced terga of the 3rd and 4th are traceable behind the larger plate. The latter bears a pair of median eyes and obsolete lateral eyes on each side. Sternal elements of prosoma almost entirely absent, traces of a prosternum and metasternum alone remaining. Rostrum free, not supported by either the prosternum or the basal segments of the appendages. Appendages of 1st pair large, chelate, bisegmented, articulated to the sides of the head-shield; appendages of 2nd pair simple, pediform, with protrusible (? suctorial) organ, and no claws at the tip; their basal segments united in the middle line and furnished with sterno-coxal process. Remaining pairs of appendages with their basal segments immovably fixed to the sternal surface, similar in form, the posterior three pairs furnished with two claws supported on long stalks; the basal segments of the 6th pair bearing five pairs of tactile sensory organs or malleoli. The prae-genital somite is suppressed. Opisthosoma composed of ten somites. Respiratory organs tracheal, opening upon the ventral surface of the 2nd and 3rd, and sometimes also of the 4th somite of the opisthosoma. A supplementary pair of tracheae opening behind the basal segment of the 4th appendage of the prosoma.
(? Intromittent organ of male lodged on the dorsal side of the 1st pair of prosomatic appendages.)
Families—Hexisopodidae (Hexisopus). Solpugidae (Solpuga, Rhagodes). Galeodidae (Galeodes). Remarks.—These most strange-looking Arachnids occur in warmer temperate, and tropical regions of Asia, Africa and America. Their anatomy has not been studied, as yet, by means of freshly-killed material, and is imperfectly known, though the presence of the coxal glands was determined by Macleod in 1884. The proportionately enormous chelae (chelicerae) of the first pair of appendages are not provided with poison glands; their bite is not venomous.
Galeodes has been made the means of a comparison between the structure of the Arachnida and Hexapod insects by Haeckel and other writers, and it was at one time suggested that there was a genetic affinity between the two groups—through Galeodes, or extinct forms similar to it. The segmentation of the prosoma and the form of the appendages bear a homoplastic similarity to the head, pro-, meso-, and meta-thorax of a Hexapod with mandibles, maxillary palps and three pairs of walking legs; while the opisthosoma agrees in form and number of somites with the abdomen of a Hexapod, and the tracheal stigmata present certain agreements in the two cases. Reference to literature (36).

|
Fig. 69.—Galeodes sp., one of the Solifugae. |
| I to VI, The six prosomatic | e, Third plate of the prosoma- |
| limbs cut short. | carrying appendage VI. |
| o, The eyes. | The prae-genital somite is |
| b, c, Demarcated areae of the | absent. |
| cephalic or first | 1, First somite of the |
| prosomatic plate | opisthosoma. |
| corresponding respectively | 2, Second do. |
| to appendages I, II, III, | S, Prosomatic tracheal |
| and to appendage IV (see | aperture between legs IV |
| fig. 68). | and V. |
| d, Second plate of the | S′ and S″, Opisthosomatic |
| prosoma-carrying | tracheal apertures. |
| appendage V. | 10, Tenth opisthosomatic |
| somite. | |
| an, Anus. |
(Original.)
Order 6. Pseudoscorpiones = Chelonethi, also called Chernetidia (see figs. 70, 71, 72).—Prosoma covered by a single dorsal shield, at most furnished with one or two diplostichous lateral eyes; sternal elements obliterated or almost obliterated. Appendages of the 1st pair bisegmented completely chelate, furnished with peculiar organs, the serrula and the lamina. Appendages of 2nd pair very large and completely chelate, their basal segments meeting in the middle line, as in the Uropygi, and provided in front with membranous lip-like processes underlying the proboscis. Appendages of the 3rd, 4th, 5th and 6th pairs similar in form and function, tipped with two claws, their basal segments in contact in the median ventral line. The prae-genital somite wide, not constricted, with large tergal plate, but with its sternal plate small or inconspicuous. Opisthosoma composed, at least in many cases, of eleven somites, the 11th somite very small, often hidden within the 10th. Respiratory organs in the form of tracheal tubes opening by a pair of stigmata in the 2nd and 3rd somites of the opisthosoma. Intromittent organ of male beneath sternum of the 1st somite of the opisthosoma.
Sub-order a. Panctenodactyli.—Dorsal plate of prosoma (carapace) narrowed in front; the appendages of the 1st pair small, much narrower, taken together, than the posterior border of the carapace. Serrula on movable digit of appendages of 1st pair fixed throughout its length, and broader at its proximal than at its distal end; the immovable digit with an external process.
- Family—Cheliferidae (Chelifer (figs. 70, 71, 72), Chiridium).
- ” Garypidae (Garypus).
(Original.)
Sub-order b. Hemictenodactyli.—Dorsal plate of prosoma scarcely narrowed in front; the appendages of the 1st pair large, not much narrower, taken together, than the posterior border of the carapace. The serrula or the movable digit free at its distal end, narrowed at the base; no external lamina on the immovable digit.
- Family—Obisiidae (Obisium, Pseudobisium).
- ” Chthoniidae (Chthonius, Tridenchthonius).
Remarks.—The book-scorpions—so called because they were, in old times, found not unfrequently in libraries—are found in rotten wood and under stones. The similarity of the form of their appendages to those of the scorpions suggests that they are a degenerate group derived from the latter, but the large size of the prae-genital somite in them would indicate a connexion with forms preceding the scorpions. Reference to literature (37).
(Original drawing by Pocock and Pickard-Cambridge.)
Order 7. Podogona = Ricinulei (see figs. 73 to 76).—Dorsal area of prosoma furnished with two shields, a larger behind representing, probably, the tergal elements of the somites, and a smaller in front, which is freely articulated to the former and folds over the appendages of the 1st pair. Ventral area without distinct sternal plates. Appendages of 1st pair, bi-segmented, completely chelate. Appendages of 2nd pair, with their basal segments uniting in the middle line below the mouth, weakly chelate at apex. Appendages of 3rd, 4th, 5th and 6th pairs similar in form; their basal segments in contact in the middle line and immovably welded, except those of the 3rd pair, which have been pushed aside so that the bases of the 2nd and 4th pairs are in contact with each other. A movable membranous joint between the prosoma and the opisthosoma, the generative aperture opening upon the ventral side of the membrane. Prae-genital somite suppressed; the opisthosma consisting of nine segments, whereof the first and second are almost suppressed and concealed within the joint between the prosoma and the opisthosoma; the following four large and manifest, and the remaining three minute and forming a slender generally-retracted tail like that of Thelyphonus. Respiratory organs tracheal, opening by a pair of spiracles in the prosoma above the base of the fifth appendage on each side. Intromittent organ of male placed at the distal end of the appendage of the 5th pair.
Family—Cryptostemmidae (Cryptostemma, Poliochera), Carboniferous.
Remarks on the Podogona.—The name given to this small but remarkable group has reference to the position of the male intromittent organ (fig. 73, e). They are small degenerate animals with a relatively firm integument. Not more than four species and twice that number of specimens are known. They have been found in West Africa and South America. A fact of special interest in regard to them is that the genus Poliochera, from the Coal Measures, appears to be a member of the same group. The name Cryptostemma, given to the first-known genus of the order, described by Guérin-Méneville, refers to the supposed concealment of the eyes by the movable cephalic sclerite. Reference to literature (38).
 |
| Fig. 76.—Cryptostemma Karschii. Extremity of the fifth pair of appendages of the female for comparison with that of the male E in fig. 73. |
Order 8. Opiliones (see fig. 77).—Dorsal area of prosoma covered by a single shield usually bearing a pair of eyes. Sternal elements much reduced. Appendages of 1st pair large, three segmented and completely chelate; of 2nd pair either simple and pediform, or prehensile and subchelate; of remaining four pairs, similar in form, ambulatory in function; the basal segment of the 2nd, 3rd and sometimes of the 4th pairs of appendages furnished with sterno-coxal (maxillary) lobe. Opisthosoma confluent throughout its breadth with the prosoma, with the dorsal plate of which its anterior tergal plates are more or less fused; at most ten opisthosomatic somites traceable; the generative aperture thrust far forwards between the basal segments of the 6th appendages. Prae-genital somite suppressed. Respiratory organs tracheal, opening by a pair of stigmata situated immediately behind the basal segments of the 6th pair of appendages on what is probably the sternum of the 2nd opisthosomatic somite and also in some cases upon the 5th segment of the legs.
Intromittent organ of male lying within the genital orifice.
Sub-order a. Laniatores.—Orifice of foetid glands opening above the coxa of the 4th appendage, not raised upon a tubercle. Orifice of coxal gland situated just behind that of the foetid gland. Sternal plate of prosoma long and narrow, with a distinct prosternal element underlying the mouth. Coxae of 4th, 5th and 6th appendages immovable. Appendages of 2nd pair, strong, usually prehensile and spiny. Genital orifice covered by an operculum.
- Families—Gonoleptidae (Gonoleptes, Goniasoma).
- Biantidae (Biantes).
- Oncopodidae (Oncopus, Pelitnus).
- Trioenonychidae (Trioenonyx, Acumontia).
Sub-order b. Palpatores.—Orifice of foetid glands opening above the coxa of the 3rd appendage, not raised upon a tubercle. Orifice of coxal gland situated between the coxae of the 5th and 6th appendages. Sternal plate of prosoma usually short and wide, rarely longer than broad; with a larger or smaller prosternal element underlying the mouth. Coxae of 3rd, 4th, 5th and 6th appendages movable or immovable. Appendages of 2nd pair weak, pediform not prehensile. Genital orifice covered by an operculum.
- Families—Phalangiidae (Phalangium, Gagrella).
- Ischyropsalidae (Ischyropsalis, Taracus).
- Nemastomidae (Nemastoma).
- Trogulidae (Trogulus, Anelasmocephalus).
Sub-order c. Cyphophthalmi (Anepignathi).—Orifice of foetid glands opening on a tubercle situated near the lateral border of the carapace above the base of the 5th appendage. Orifice of coxal gland probably situated at base of coxa of 5th appendage; sternal plate of prosema minute or absent; no prosternal element underlying the mouth. Coxae of 5th and 6th, and usually also of 4th appendages immovable. Appendages of 2nd pair weak, pediform, not prehensile. Genital orifice not covered by an operculum.
- Families—Sironidae (Siro, Pettalus).
- Stylocellidae (Stylocellus).
Remarks on the Opiliones.—These include the harvest-men, sometimes called also daddy-long-legs, with round undivided bodies and very long, easily-detached legs. The intromittent organs of the male are remarkable for their complexity and elaboration. The confluence of the regions of the body and the dislocation of apertures from their typical position are results of degeneration. The Opiliones seem to lead on from the Spiders to the Mites. Reference to literature (39).
Apparently related to the Opiliones are two extinct groups, the Anthracomarti and Phalangiotarbi, which are not known to have survived the Carboniferous period. In the Anthracomarti the opisthosoma was movably articulated to the prosoma, and consisted of from eight to ten segments furnished with movable lateral plates, the anal segment being overlapped dorsally by a laminate expansion of the preceding segment. The carapace of the prosoma was unsegmented and often bore a pair of eyes. The appendages of the 2nd pair were slender and pediform; those of the 3rd, 4th, 5th and 6th pairs were similar in form and ambulatory in function with their basal segments arranged round a sternal area as in the order Araneae. The best-known genera were Anthracomartus and Eophognus.
In the Phalangiotarbi the appendages resembled those of the Anthracomarti, except that the basal segments of the last four pairs were usually approximated in the middle line leaving a long and narrow sternal area between; and the carapace of the prosoma was unsegmented. The prosoma and opisthosoma were broadly confluent and probably immovably welded together. The opisthosoma consisted of eight or nine segments, whereof the anterior five or six were very short in the dorsal region, and the posterior three exceptionally large with the anal orifice terminal.
Several genera have been established, the best-characterized being Geraphognus and Architarbus.
Order 9. Rhynchostomi = Acari (see fig. 78).—Degenerate Arachnids resembling the Opiliones in many structural points, but chiefly distinguishable from them by the following features:—The basal segments of the appendages of the 2nd pair are united in the middle line behind the mouth, those of the 3rd, 4th, 5th and 6th pairs are widely separated and not provided with sterno-coxal (maxillary) lobes, and take no share in mastication; the respiratory stigmata, when present, belong to the prosoma, and the primitive segmentation of the opisthosoma has entirely or almost entirely disappeared.
Sub-order a. Notostigmata.—Opisthosoma consisting of ten segments defined by integumental grooves, each of the anterior four of these furnished with a single pair of dorsally-placed spiracles or tracheal stigmata.
- Family—Opilioacaridae (Opilioacarus).
Sub-order b. Cryptostigmata.—Integument hard, strengthened by a continuously chitinized dorsal and ventral sclerite. Tracheae typically opening by stigmata situated in the articular sockets (acetabula) of the 3rd, 4th, 5th and 6th pairs of appendages.
- Family—Oribatidae (Oribata, Nothrus, Hoplophora).
Sub-order c. Metastigmata.—Integument mostly like that of the Cryptostigmata. Tracheae opening by a pair of stigmata situated above and behind the base of the 4th or 5th or 6th pair of appendages.
- Families—Gamasidae (Gamasus, Pteroptus).
- Argasidae (Argas, Ornithodoros).
- Ixodidae (Ixodes, Rhipicephalus).
Sub-order d. Prostigmata.—Integument soft, strengthened by special sclerites, those on the ventral surface of the prosoma apparently representing the basal segments of the legs embedded in the skin. Tracheae, except in the aquatic species in which they are atrophied, opening by a pair of stigmata situated close to or above the base of the appendages of the 1st pair (mandibles).
- Families—Trombidiidae (Trombidium, Tetranychus).
- Hydrachnidae (Hydrachna, Atax).
- Halacaridae (Halacarus, Leptognathus).
- Bdellidae (Bdella, Eupodes).
Sub-order e. Astigmata.—Degenerate, mostly parasitic forms approaching the Prostigmata in the development of integumental sclerites and the softness of the skin, but with the respiratory system absent.
- Families—Tyroglyphidae (Tyroglyphus, Rhizoglyphus).
- Sarcoptidae (Sarcoptes, Analges).
Sub-order f. Vermiformia.—Degenerate atracheate parasitic forms with the body produced posteriorly into an annulated caudal prolongation, and the 3rd, 4th, 5th and 6th pairs of appendages short and only three-jointed.
- Family—Demodicidae (Demodex).
Sub-order g. Tetrapoda.—Degenerate atracheate gall-mites in which the body is produced posteriorly and annulated, as in Demodex, but in which the appendages of the 3rd and 4th pairs are long and normally segmented and those of the 5th and 6th pairs entirely absent.
- Family—Eriophyidae (Eriophyes, Phyllocoptes).
Remarks on the Rhynchostomi.—The Acari include a number of forms which are of importance and special interest on account of their parasitic habits. The ticks (Ixodes) are not only injurious as blood-suckers, but are now credited with carrying the germs of Texas cattle-fever, just as mosquitoes carry those of malaria. The itch-insect (Sarcoptes scabiei) is a well-known human parasite, so minute that it was not discovered until the end of the 18th century, and “the itch” was treated medicinally as a rash. The female burrows in the epidermis much as the female trap-door spider burrows in turf in order to make a nest in which to rear her young. The male does not burrow, but wanders freely on the surface of the skin. Demodex folliculorum is also a common parasite of the sebaceous glands of the skin of the face in man, and is frequent in the skin of the dog. Many Acari are parasitic on marine and freshwater molluscs, and others are found on the feathers of birds and the hair of mammals. Others have a special faculty of consuming dry, powdery vegetable and animal refuse, and are liable to multiply in manufactured products of this nature, such as mouldy cheese. A species of Acarus is recorded as infesting a store of powdered strychnine and feeding on that drug, so poisonous to larger organisms. Reference to literature (40).
Authorities cited by numbers in the text.—1. Strauss-Dürckheim (as reported by MM. Riester and Sanson in an appendix to the sixth volume of the French translation of Meckel’s Anatomy, 1829); 2. Lankester, “Limulus an Arachnid,” Quart. Journ. Micr. Sci. vol. xxi. N.S., 1881; 3. Idem, “On the Skeletotrophic Tissues of Limulus, Scorpio and Mygale,” Quart. Journ. Micr. Sci. vol. xxiv. N.S., 1884; 4. Idem. Trans. Zool. Soc. vol. xi., 1883; 5. Lankester and A. G. Bourne, “Eyes of Limulus and Scorpio,” Quart. Journ. Micr. Sci. vol. xxiii. N.S., Jan. 1883; 6. Milne-Edwards, A., “Recherches sur l’anatomie des Limules,” Ann. Sci. Nat. 5th Series, Zoologie, vol. xvii., 1873; 7. Owen, Richard, “Anatomy of the King-Crab,” Trans. Linn. Soc. Lond., vol. xxviii., 1872; 8. Kishinouye, “Development of Limulus longispina,” Journal of the Science College of Japan, vol. v., 1892; 9. Brauer, “Development of Scorpion,” Zeitschrift für wiss. Zoologie, vol. lix., 1895; 10. Hansen, H. J., “Organs and Characters in Different Orders of Arachnida,” Entomol. Meddel. vol. iv. pp. 137-149; 11. Watase, “On the Morphology of the Compound Eyes of Arthropods,” Studies from the Biolog. Lab. Johns Hopkins University, vol. iv. pp 287-334; 12. Newport, George, “Nervous and Circulatory Systems in Myriapoda and Macrourous Arachnids,” Phil. Trans. Roy. Soc., 1843; 13. Lankester, “Coxal Glands of Limulus, Scorpio and Mygale,” Quart. Journ. Micr. Sci. vol. xxiv. N.S., 1884; 13a. W. Patten and A. P. Hazen, “Development of the Coxal Glands of Limulus,” Journ. of Morphology, vol. xvi., 1900; 13b. Bernard, “Coxal Glands of Scorpio,” Ann. and Mag. Nat. Hist. vol. xii., 1893, p. 55; 14. Benham, “Testis of Limulus,” Trans. Linn. Soc., 1882; 15. Lankester, “Mobility of the Spermatozoa of Limulus,” Quart. Journ. Micr. Sci. vol. xviii. N.S., 1878; 16. Korschelt and Heider, Entwickelungsgeschichte (Jena, 1892), ibique citata; 17. Laurie, M., “The Embryology of a Scorpion,” Quart. Journ. Micr. Sci. vol. xxxi. N.S., 1890, and “On Development of Scorpio fulvipes,” ibid. vol. xxxii., 1891; 18. Lankester (Homoplasy and Homogeny), “On the Use of the term Homology in Modern Zoology,” Ann. and Mag. Nat. Hist., 1870; 19. Idem, “Degeneration, a Chapter in Darwinism,” 1878, reprinted in the Advancement of Science (Macmillan, 1890); 20. Idem, “Limulus an Arachnid,” Q. J. Micr. Sci. vol. xxi. N.S.; 21. Claus, “Degeneration of the Acari and Classification of Arthropoda,” Anzeiger d. k. k. Akad. Wissen. Wien, 1885; see also Ann. and Mag. Nat. Hist. (5) vol. xvii., 1886, p. 364, and vol. xix. p. 225; 22. Lindström, G., “Researches on the Visual Organs of the Trilobites,” K. Svenska Vet. Akad. Handl. xxxiv. No. 8, pp. 1-86, Pls. i.-vi., 1901; 22*. Zittel, American edition of his Palaeontology (the Macmillan Co., New York), where ample references to the literature of Trilobitae and Eurypteridae will be found; also references to literature of fossil Scorpions and Spiders; 23. Hoek, “Report on the Pycnogonida,” Challenger Expedition Reports, 1881; Meinert, “Pycnogonida of the Danish Ingolf Expedition,” vol. iii., 1899; Morgan, “Embryology and Phylogeny of the Pycnogonids,” Biol. Lab. Baltimore, vol. v., 1891; 24. Bourne, A. G., “The Reputed Suicide of the Scorpion,” Proc. Roy. Soc. vol. xlii. pp. 17-22; 25. Lankester, “Notes on some Habits of Scorpions,” Journ. Linn. Soc. Zool. vol. xvi. p. 455, 1882; 26. Huxley, “Pharynx of Scorpion,” Quart. Journ. Micr. Sci. vol. viii. (old series), 1860, p. 250; 27. Pocock, “How and Why Scorpions hiss,” Natural Science, vol. ix., 1896; cf. idem, “Stridulating Organs of Spiders,” Ann. and Mag. Nat. Hist. (6), xvi. pp. 230-233; 28. Kraepelin, Das Thierreich (Scorpiones et Pedipalpi) (Berlin, 1899); Peters, “Eine neue Eintheilung der Skorpione,” Mon. Akad. Wiss. Berlin, 1861; Pocock, “Classification of Scorpions,” Ann. and Mag. Nat. Hist. (6) xii., 1893; Thorell and Lindström, “On a Silurian Scorpion,” Köngl. Svens. Vet. Akad. Handl. xxi. No. 9, 1885; 29. Cambridge, O. P., “A New Family (Tartarides) and Genus of Thelyphonidea,” Ann. and Mag. Nat. Hist. (4) x., 1872, p. 413; Cook, “Hubbardia, a New Genus of Pedipalpi,” Proc. Entom. Soc. Washington, vol. iv., 1899; Thorell, “Tartarides, &c.” Ann. Mus. Genova, vol. xxvii., 1889; 30. M'Cook, American Spiders and their Spinning Work (3 vols.; Philadelphia, 1889–1893); 31. Peckham, “On Sexual Selection in Spiders,” Occasional Papers Nat. Hist. Soc. Wisconsin, vol. i. pp. 1-113, 1889; 32. Moggridge, Harvesting Ants and Trap-Door Spiders (1873); 33. Bertkau, Ph., Arch. f. Naturgesch. vol. xlviii. pp. 316-362; Idem, same journal, 1875, p. 235, and 1878, p. 351; Cambridge, O. P., “Araneidea” in Biologia Centr. Americana, vols. i. and ii. (London, 1899); Keyserling, Spinnen Amerikas (Nuremberg, 1880–1892); Pocock, “Liphistius and the Classification of Spiders,” Ann. and Mag. Nat. Hist. (6) x., 1892; Simon, Hist. nat. des Araignées, vols. i. and ii., 1892, 1897; Wagner, “L’Industrie des Araneína,” Mém. Acad. St-Pétersbourg; Idem, “La Mue des Araignées,” Ann. Sci. Nat. vol. vi.; 34. Grassi, G. B. “Intorno ad un nuovo Aracnide artrogastro (Koenenia mirabilis) &c.” Boll. Soc. Ent. Ital. vol. xviii., 1886; 35. H. J. Hansen and Sörensen, “The Order Palpigradi, Thorell (Koenenia), and its Relationships with other Arachnida,” Ent. Tidskr. vol. xviii. pp. 233-240, 1898; Kraepelin, Das Thierreich (Berlin, 1901); 36. Bernard. “Compar. Morphol. of the Galeodidae,” Trans. Linn. Soc. Zool. vol. vi., 1896, ibique citata; Dufour, “Galeodes,” Mém. prés. Acad. Sci. Paris, vol. xvii., 1862; Kraepelin, Das Thierreich (Berlin, 1901); Pocock, “Taxonomy of Solifugae,” Ann. and Mag. Nat. Hist. vol. xx.; 37. Balzan, “Voyage au Vénézuela (Pseudoscorpiones),” Ann. Soc. Entom. France, 1891, pp. 497-522; 38. Guérin-Méneville, Rev. Zool., 1838, p. 11; Karsch, “Ueber Cryptostemma Guer.” Berliner entom. Zeitschrift, xxxviii. pp. 25-32, 1892; Thorell, “On an apparently new Arachnid belonging to the family Cryptostemmidae,” Westv. Bihang Svenska Vet. Akad. Handligar, vol. xvii. No. 9, 1892; 39. Hansen and Sörensen, On Two Orders of Arachnida (Cambridge, 1904); Sörensen, “Opiliones laniatores,” Nat. Tidskr. (3) vol. xiv., 1884; Thorell, “Opilioni,” Ann. Mus. Genova, vol. viii., 1876; 40. Berlese, “Acari, &c., in Italia reperta” (Padova, 1892); Canestrini, Acarofauna Italiana (Padova, 1885); Canestrini and Kramer, “Demodicidae and Sarcoptidae” in Das Thierreich (Berlin, 1899); Michael, “British Oribatidae,” Ray Soc.; Idem, “Oribatidae” in Das Thierreich (Berlin, 1898); Idem, “Progress and Present State of Knowledge of Acari,” Journ. Roy. Micr. Soc., 1894; Nalepa, “Phytoptidae,” Das Thierreich (Berlin, 1898); Trouessart, “Classification des Acariens,” Rev. Sci. Nat. de l’ouest. p. 289, 1892; Wagner, Embryonal Entwick, von Ixodes (St Petersburg, 1893); 41. Bertkau, Ph., “Coxaldrusen der Arachniden,” Sitzb. Niederl. Gesellsch., 1885; 42. Patten, W., “Brain and Sense Organs of Limulus,” Quart. Journ. Mic. Sci. vol. xxxv., 1894; see also his “Origin of Vertebrates from Arachnids,” ibid. vol. xxxi.
Authorities not cited by numbers in the text:—
Lung-books:—Berteaux, “Le Poumon des Arachnides,” La Cellule, vol. v. 1891; Jawarowski, “Die Entwick. d. sogen. Lunge bei der Arachniden,” Zeitsch. wiss. Zool. vol. lviii., 1894; Macleod, “Recherches sur la structure et la signification de l’appareil respiratoire des Arachnides,” Arch. d. Biologie. vol. v., 1884; Schneider, A., “Mélanges arachnologiques,” in Tablettes zoologiques, vol. ii. p. 135, 1892; Simmons, “Development of Lung in Spiders,” Amer. Journ. Science, vol. xlviii., 1894. Coxal Glands:—Bertkau, “Ueber die Coxaldrusen der Arachniden,” Sitzb. d. Niederl. Gesellsch., 1885; Loman, “Altes und neues über das Nephridium (die Coxaldrüse) der Arachniden,” Bÿd. tot de Dierkunde, vol. xiv., 1887; Macleod, “Glande coxale chez les Galéodes,” Bull. Acad. Belg. (3) vol. viii., 1884; Pelseneer, “On the Coxal Glands of Mygale,” Proc. Zool. Soc., 1885; Tower, “The External Opening of the brick-red Glands of Limulus,” Zool. Anzeiger, vol. xviii. p. 471, 1895. Ento-sternite:—Schimkewitsch, “Bau und Entwick. des Endosternites der Arachniden,” Zool. Jahrb., Anal. Abtheil., vol. viii., 1894. Embryology:—Balfour, “Development of the Araneina,” Q. J. Micr. Sci. vol. xx., 1880; Kingsley, “The Embryology of Limulus,” Journ. Morphology, vols. vii. and viii.; Kishinouye, “Development of Araneina,” Journ. Coll. Sci. Univ. of Japan, vol. iv., 1890; Locy, “Development of Agelena,” Bull. Mus. Harvard, vol. xii., 1885; Metchnikoff, “Embryologie d. Scorpion,” Zeit. wiss. Zool. vol. xxi., 1871; Idem, “Embryol. Chelifer,” Zeit. wiss. Zool. vol. xxi., 1871; Schimkewitsch, “Développement des Araignées,” Archives d. Biologie, vol. vi. 1887. Sense organs:—Bertkau, “Sinnesorgane der Spinnen,” Arch. f. mikros. Anat. vol. xxvii. p. 589, 1886; Graber, “Unicorneale Tracheaten Auge,” Arch. f. mikr. Anat. vol. xvii., 1879; Grenacher, Gehörorgane der Arthropoden (Göttingen, 1879); Kishinouye, “Lateral Eyes of Spiders,” Zool. Anz. vol. xiv. p. 381, 1891; Purcell, “Phalangiden Augen,” Zool. Anzeiger, vol. xv. p. 461.
General works on Arachnida:—Blanchard, “Les Arachnides” in L’Organisation du regne animal; Gaubert, “Recherches sur les Arachnides,” Ann. Sci. Nat. (7) vol. xiii., 1892; Koch, C., Die Arachniden (16 vols., Nuremberg, 1831–1848); Koch, Keyserling and Sörensen, Die Arachniden Australiens (Nuremberg, 1871–1890); Pocock, Arachnida of British India (London, 1900); Idem, “On African Arachnida,” in Proc. Zool. Soc. and Ann. and Mag. Nat. Hist., 1897–1900; Simon, Les Arachnides de la France (7 vols., Paris, 1874–1881); Thorell, “Arachnida from the Oriental Region,” Ann. Mus. Genova, 1877–1899. (E. R. L.)
- ↑ See the article Arthropoda for the use of the term “prosthomere.”
- ↑ See fig. 12 in the article Arthropoda.
- ↑ Though ten is the prevailing number of retinula cells and rhabdomeres in the lateral eye of Limulus, Watase states that they may be as few as nine and as many as eighteen.
- ↑ A great deal of superfluous hypothesis has lately been put forward in the name of “the principle of convergence of characters” by a certain school of palaeontologists. The horse is supposed by these writers to have originated by separate lines of descent in the Old World and the New, from five-toed ancestors! And the important consequences following from the demonstration of the identity in structure of Limulus and Scorpio are evaded by arbitrary and even phantastic invocations of a mysterious transcendental force which brings about “convergence” irrespective of heredity and selection. Morphology becomes a farce when such assumptions are made. (E. R. L.)
- ↑ A pair of round tubercles on the labram (camerostome or hypostoma) of several species of Trilobites has been described and held to be a pair of eyes (22). Sense-organs in a similar position were discovered in Limulus by Patten (42) in 1894.
- ↑ The writer is indebted to R. I. Pocock, assistant in the Natural History departments of the British Museum, for valuable assistance in the preparation of this article and for the classification and definition of the groups of Eu-arachnida here given. The general scheme and some of the details have been brought by the writer into agreement with the views maintained in this article. Pocock accepts those views in all essential points and has, as a special student of the Arachnida, given to them valuable expansion and confirmation. The writer also desires to express his thanks to Messrs. Macmillan & Co. for permission to use figs. 22, 43, 44 and 45, which are taken from Parker and Haswell’s Text-book of Zoology; and to Messrs. Swan Sonnenschein & Co. for the loan of several figures from the translations published by them of the admirable treatise on Embryology by Professors Korschelt and Heider; also to the publishers of the treatise on Palaeontology by Professor Zittel, Herr Oldenbourg and The Macmillan Co., New York, for several cuts of extinct forms.
- ↑ Pocock suggests that the area marked vii. in the outline figure of the dorsal view of Limulus (fig. 7) may be the tergum of the suppressed prae-genital somite. Embryological evidence must settle whether this is so or not.