1911 Encyclopædia Britannica/Endospora
ENDOSPORA, a natural group or class of the Sporozoa, consisting of the orders Myxosporidia, Actinomyxidia, Sarcosporidia and Haplosporidia, together with various insufficiently-known forms (Sero- and Exosporidia), regarded at present as Sporozoa incertae sedis. The distinguishing feature of the group is that the spore-mother-cells (pansporoblasts) arise in the interior of the body of the parent-individual; in other words, sporulation is endogenous. Another very general character—though not so universal—is that the adult trophozoite possesses more than one nucleus, usually many (i.e. it is multinucleate). In the majority of forms, though apparently not in all (e.g. certain Microsporidia), sporulation goes on coincidently with growth and trophic life. With regard to the origin of the group, the probability is greatly in favour of a Rhizopod ancestry. The entire absence, at any known period, of a flagellate or even gregariniform phase; on the other hand, the amoeboid nature of the trophozoites in very many cases together with the formation of pseudopodia; and, lastly, the simple endogenous spore-formation characteristic of the primitive forms,—are all points which support this view, and exclude any hypothesis of a Flagellate origin, such as, on the contrary, is probably the case in the Ectospora (q.v.).
1. Order Myxosporidia. The Myxosporidia, or, more correctly, the dense masses formed by their spores, were well known to the earlier zoological observers. The parasites in fishes were called by Müller “fish-psorosperms,” a name which has stuck to them ever since, although, as is evident from the meaning of the term (“mange-seed”), Müller had little idea of the true nature of the bodies. Other examples, infesting silkworms, have also long been known as “Pèbrine-corpuscles,” from the ravaging disease which they produce in those caterpillars in France, in connexion with which Pasteur did such valuable work. The foundation of our present morphological and biological knowledge of the order was well laid by the admirable researches of Thèlohan in 1895. In spite, however, of the contributions of numerous workers since then (e.g. Doflein, Cohn, Stempell and others), there are still one or two very important points, such as the occurrence of sexual conjugation, upon which light is required.
Although pre-eminently parasites of fishes, Myxosporidia also occur, in a few cases, in other Vertebrates (frogs and reptiles); no instance of their presence in a warm-blooded Vertebrate has, however, yet been described. One suborder (the Microsporidia or Cryptocystes) is pretty Occurrence and habitat. equally distributed between fishes on the one hand and Invertebrates—chiefly, but not exclusively, Arthropods—on the other. The parasites are frequently the cause of severe and fatal illness in their hosts, and devastating epidemics of myxosporidiosis have often been reported (e.g. among carp and barbel in continental rivers, due to a Myxobolus, and among crayfish in France, to Thelohania).
The seat of the invasion and the mode of parasitism are extremely varied. Practically any organ or tissue may be attacked, excepting, apparently, the testis and cartilage and bone. In one instance at least (that of Nosema bombycis of the silkworm) the parasites penetrate into the ova, so that true hereditary infection occurs, the progeny being born with the disease. The parasites may be either free in some lumen, such as that of the gall bladder or urinary bladder (not of the alimentary canal, or the body-cavity itself), when they are known as coelozoic forms; or in intimate relation with some tissue, intracellular while young but becoming intercellular in the adult phase (histozoic forms); or entirely intracellular (cytozoic forms). Among the histozoic and cytozoic types, moreover, two well-defined conditions, concentration and diffuse infiltration, occur. In the former, the parasitic zone is strictly limited, and well-marked cysts are formed; in the latter, the infection spreads throughout the neighbouring tissue, and the parasitic development becomes inextricably commingled with the host’s cells. Sometimes, as shown by Woodcock (45), there may be an attempt on the part of the host’s tissue to circumscribe and check the growth of these parasitic areas, which results in the formation of pseudocysts, quite different in character from true cysts.
The most noticeable feature about the Myxosporidian trophozoite is its amoeboid and Rhizopod-like character. Pseudopodia of various kinds, from long slender ones (fig. 3, B) to short blunt lobose ones, are of general occurrence, being most easily observed, of Morphology. course, in the free-living forms. The pseudopodia serve chiefly for movement and attachment, and never, it should be noted, for the injection of solid food-particles, as in the case of Amoebae. The general protoplasm is divisible into ectoplasm and endoplasm. The former is a clear, finely-granular layer, of which the pseudopodia are mainly constituted (fig. 3, A). In one or two instances (e.g. Myxidium lieberkühnii) the ectoplasm shows a vertical striation, and in the older trophozoites breaks down partially, appearing like a fur of delicate, non-motile filaments. A somewhat similar modification is found in Myxocystis. The endoplasm is more fluid, and contains numerous inclusions of a granular nature, as well as vacuoles of varying size. In the endoplasm are lodged the nuclei, of which in an adult trophozoite there may be very many; they are all derived by multiplication from the single nucleus with which the young individuals begin life, the number increasing as growth proceeds.

|
 |
|
From Wasielewski, Sporozoenkunde. | |
|
Fig. 3.—A. Trophozoite of Sphaerospora divergens, Thél. (par. Blennius and Crenilabrus), × 750. ec, Ectoplasm; en, endoplasm; sp, spores, each with four pole capsules. | |
|
From Lankester’s Treatise on Zoology, vol. Protozoa. | |
|
B. Spore-bearing trophozoite of Leptotheca agilis, Thél. (par. Trygon and Scorpaena), × 750. ps, Pseudopodia localized at the anterior end; f.gr, fatty granules similarly localized; r.gr, refringent granules; sp, spores, two in number. | |
Spore-formation goes on entirely in the endoplasm. The number of spores formed is very variable. It may be as low as two (as in free-living forms, e.g. Leptotheca), in which case a large amount of trophic protoplasm is unconverted Spore-formation; multipli-cative processes. into spores; or, on the other hand, the number of spores may be very great (as in tissue-parasites), practically the whole of the parent-body being thus used up. The sporont may or may not encyst at the commencement of sporulation. In the free-living forms there is no cyst-membrane secreted; but in certain Glugeidae, on the other hand, the ectoplasm becomes altered into a firm, enclosing layer, the ectorind, which forms a thick cyst-wall (fig. 5). The process of sporulation begins by the segregation of small quantities of endoplasm around certain of the nuclei, to form little, rounded bodies, the pansporoblasts. There may be either very many or only few pansporoblasts developed; in some cases, indeed, there is only one, the sporont either itself becoming a pansporoblast (certain Microsporidia), or giving rise to a solitary one (Ceratomyxidae). The pansporoblast constituted, nuclear multiplication goes on preparatory to the formation of sporoblasts, which in their turn become spores (see figs. 4 and 5). Not all the nuclei thus formed, however, are made use of. In the Phaenocystes there are always two sporoblasts developed in each pansporoblast; in the Cryptocystes there may be from one to several. Around each sporoblast a spore-membrane is secreted, which usually has the form of two valves. It has recently been shown by Léger and Hesse (29b) that, in many Phaenocystes at any rate, each of these valves is formed by a definite nucleated portion of the sporoblast.
The spores themselves vary greatly in size and shape (figs. 7 and 8). They may be as small as 1.5 μ by 1 μ (as in a species of Nosema), or as large as 100 μ by 12 μ (as in Ceratomyxa). A conspicuous feature in the structure of a fully-developed spore is the polar-capsules, of which there may be either 1, 2, or 4 to each. In the Phaenocystes the polar-capsules are visible in the fresh condition, but not in the Cryptocystes. The polar-capsule is an organella which recalls the nematocyst of a Hydrozoan, containing a spirally-coiled filament, often of great length, which is shot out on the application of a suitable stimulus. Normally, as was ingeniously shown by Thélohan (43), the digestive juices of the fresh host serve this purpose, but various artificial means may suffice. The function of the everted filament is probably to secure the attachment of the spore to the epithelium of the new host. In the Phaenocystes, in connexion with each polar-capsule, a small nuclear body can be generally made out; these two little nuclei are those of the two “capsulogenous” areas of the protoplasm of the pansporoblast, which formed the capsules. The sporoplasm, representing the sporozoite, is always single. Nevertheless, in the Phaenocystes it is invariably binuclear; and, in the Microsporidia, the nucleus, at first single, gives rise later to four nuclei, two of which are regarded by Stempell (42) as corresponding to those of two polar-capsules (of which only one is developed in the spore), the remaining two representing germ-nuclei. Hence it is possible that the Myxosporidian sporoplasm really consists of two, incompletely-divided (sister) germs. Moreover, it is supposed by some that these two nuclei fuse together later, this act representing a sexual conjugation; since the earliest known phases of young trophozoites (amoebulae) have been described as uninuclear.

| |
| From Lankester’s Treatise on Zoology, vol. Protozoa, after Thélohan. | |
|
Fig. 4.—Stages in spore-formation. All the figures are from Myxobolus ellipsoides, except a and f, which are from M. pfeifferi. a, Differentiation of the pansporoblast (p.sp). b, Pansporoblast with two nuclei. c and d, Pansporoblasts with six and ten nuclei respectively; in d, four of the nuclei are degenerating. e, Pansporoblast segmented into two definitive sporoblasts, each with three nuclei. In the next four figures the definitive sporoblast, or the spore produced from it, is alone figured. f, Definitive sporoblast segmented into three masses, the capsulogenous cells (c.g.c) and the sporoplasm (sp.p), within an envelope, the spore membrane (sp.m). g, More advanced stage. h, Spore completely developed, with two polar capsules and sporoplasm containing an iodinophilous vacuole. i, Abnormal spore containing six polar capsules. | |
| n, Nuclei. | sp.bl, Definitive sporoblast. |
| r.n, Residuary nuclei. | vac, Vacuole. |
| r.p.c, Rudiment of p.c, polar capsule. | n.p.c, Nuclei of polar capsules. |
| iod.vac, Iodinophilous vacuole. | n.sp, Nuclei of sporoplasm. |

|
| From Woodcock, Proc. and Trans. of the Liverpool Biological Society, 1904. |
|
Fig. 5.—Part of the periphery of a cyst of Glugea stephani, in the intestinal wall of the plaice, showing sporoblast and spore-formation. ect, Ectorind. end, Endoplasm. endoth, Fold of the mucous membrane, normal in character. p.sp.bl, Various stages in the development of the pansporoblasts. sp, Ripe spores, filling the greater part of the cyst. n, Large (vegetative) nuclei. |

|
|
From Lankester’s Treatise on |
Fig. 6.—Formation of buds by multiple plasmotomy in Myxidium lieberkühnii, Bütschli (par. Esox and Lota) after Cohn. |
In addition to spore-formation, two or three modes of endogenous reproduction, serving for auto-infection, have been made known. One, termed by Doflein plasmotomy, consists either in the division of the (multinucleate) trophozoite into two, by more or less equal fission (simple plasmotomy), or in the budding-off, from the parent trophozoite, of several portions (example: Myxidium lieberkühnii, fig. 6). A variety of this method has been described by Stempell (40) in the case of the young trophozoites (meronts) of Thelohania mülleri, which may divide into two while still uninuclear; and by rapid successive divisions chains of meronts may be formed, the different individuals being incompletely separated. Another method, which is probably chiefly responsible for the rapid spread of tissue-parasites and cell-parasites (such as Myxobolidae and Glugeidae) through their host’s tissue in the condition of diffuse infiltration, consists in multiple nuclear division, and the liberation of amoebulae while the parasite is yet quite young and possesses only few nuclei. As Woodcock has pointed out in considering the case of Glugea stephani, it is very probable that this “multiplicative reproduction,” in diffuse infiltration, is to be looked upon as a separation of the pansporoblast-rudiments as daughter-individuals; i.e. that the pansporoblasts are, in certain circumstances, capable of independent existence as little sporonts. A further stage in this direction of evolution is seen, according to Stempell, in Thelohania, Pleistophora and other types where the whole individual becomes one reproductive organella; such forms are to be considered as examples of a phylogenetic individualization of the pansporoblasts, which now exist as solitary sporonts. An extreme case of this “reduction of the individual” is found, apparently in the genus Nosema, as lately characterized by Perez (34), where vast numbers of minute entirely independent sporonts (pansporoblasts) are produced, each of which gives rise to only a single spore.
The Myxosporidia are divided into two suborders, the Phaenocystes and the Cryptocystes. Some authors have of late years separated these two divisions and raised each to the rank of a distinct order, considering that they are not more closely related to each other than to other Endosporan orders. We think this is a mistake; and it is very interesting to find that Léger and Hesse (1908) have described (29a) a new genus of Phaenocystes, Coccomyxa, which represents a type intermediate between these two suborders, and shows that they are closely connected.
Suborder 1: Phaenocystes, Gurley. Spores relatively large, with generally two or four polar-capsules, visible in the fresh condition. There are nearly always two spores formed Classification. in each pansporoblast.
Section (a): Disporea. Only two spores (i.e. one pansporoblast) produced in each individual trophozoite. The greatest length of the spore is at right angles to the plane of the suture.
One family, Ceratomyxidae, including two genera, Ceratomyxa (fig. 3, B) and Leptotheca, typically “free” parasites, mostly from the gall bladders of fishes. The valves of the spore in the former genus are prolonged into hollow cones. The type-species of this genus is C. sphaerulosa, from Mustelus and Galeus; that of Leptotheca is L. agilis, from Trygon.
Section (b): Polysporea. More than two spores, generally very many, are produced typically by each individual trophozoite. The greatest length of the spore is usually in the sutural plane.
Family, Myxidiidae. Spores with two polar-capsules, and without an iodinophilous vacuole in the sporoplasm. Mostly “free” parasites. Gen. Sphaerospora. Four or five species are known, from the kidneys or gall bladder of fishes (fig. 3, A). One, S. elegans, is interesting in that it affords a transition between the two sections, being disporous. Gen. Myxidium; spores elongated and fusiform, with a polar capsule at each extremity. The best-known species is M. lieberkühnii, from the urinary bladder of the pike. One or two species occur in reptiles. Other genera are Sphaeromyxa, Cystodiscus, Myxosoma and Myxoproteus.
Family, Chloromyxidae. Spores with four polar capsules and no iodinophilous vacuole. One genus, Chloromyxum, of which several species are known; the type being C. leydigi, from the gall bladder of various Elasmobranchs (fig. 7, B).

|
|
Fig. 7.—A. Spore of Ceratomyxa sphaerulosa, Thél. (par. Mustelus |
|
B. Spores of Chloromyxidae, after Thélohan. a, Chloromyxum |
|
C. Spores of Myxobolus ellipsoides, Thél. The spores on the left |
Family, Myxobolidae. Spores with two polar-capsules (exceptionally one), and with a characteristic iodinophilous vacuole in the sporoplasm. Typically tissue parasites of Teleosteans, often very dangerous. Genus Myxobolus. Spores oval or rounded, without a tail-like process. Very many species are known, which are grouped into three subsections: (a) forms with only one polar-capsule, such as M. piriformis, of the tench; (b) forms with two unequal capsules, e.g. M. dispar from Cyprinus and Leuciscus; and (c) the great majority of species with two equal polar-capsules, including M. mülleri, the type-species, from different fish, M. cyprini and M. pfeifferi, the cause of deadly disease in carp and barbel respectively and others. Other genera are Henneguya and Hoferellus, differing from Myxobolus in having, respectively, one or two tail-like processes to the spore. Lentospora, according to Plehn (37), lacks an iodinophilous vacuole.
Family Coccomyxidae. The pansporoblasts produce (probably) only one spore. Spore oval, large (14 μ by 5.5 μ), with a single very large polar-capsule. Sporoplasm with no vacuole. Single genus Coccomyxa, with the characters of the family. One species, C. morovi, Léger and Hesse, from the gall bladder of the sardine. The spore greatly resembles a Cryptocystid spore.
Suborder 2: Cryptocystes, Gurley (= Microsporidia, Balbiani). Spores minute, usually pear-shaped, with only one polar-capsule, which is visible only after treatment with reagents. The number of spores formed in each pansporoblast varies greatly in different forms.
Section (a): Polysporogenea. The trophozoite produces numerous pansporoblasts, each of which gives rise to many spores. Genus Glugea, with numerous species, of which the best-known is G. anomala, from the stickleback (fig. 1). The genus Myxocystis, which has been shown by Hesse (24) to be a true Microsporidian, is placed by Perez in this section, but this is a little premature, as Hesse does not describe the exact character of the sporulation, i.e. with regard to the number of pansporoblasts and the spores they produce.
Section (b): Oligosporogenea. The trophozoite becomes itself the (single) pansporoblast. In Pleistophora, the pansporoblast produces many spores; P. typicalis, from the muscles of various fishes (fig. 2), is the type-species. In Thelohania, eight spores are formed; the different species are parasitic in Crustacea. In Gurleya, parasitic in Daphnia, only four are formed; and, lastly, in Nosema (exs. N. pulvis, from Carcinus, and, most likely, N. bombycis, of the silkworm), each pansporoblast produces only a single spore.
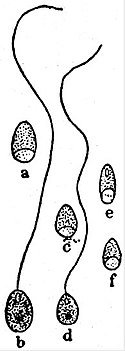
|
|
From Lankester’s Treatise |
|
Fig. 8.—Spores of a and b, Pleistophora c and d, Thelohania e, Glugea depressa, f, G. acuta, Thél. |
2. Order—Actinomyxidia. This order comprises a peculiar group of parasites, first described by A. Stolc in 1899, which are restricted to Oligochaete worms of the family Tubificidae. Most forms attack the intestinal wall, often destroying its epithelium over considerable areas; but one genus, Sphaeractinomyxon, inhabits the body-cavity of its host. The researches of Caullery and Mesnil (10-12) and of Léger (28 and 29) have shown that the parasites exhibit the typical features of the Endospora, and the spores possess the characteristic polar-capsules of the Myxosporidian spore, but differ therefrom by their more complicated structure.
The growth and development of an Actinomyxidian have been recently worked out by Caullery and Mesnil in the case of Sphaeractinomyxon stolci. A noteworthy point is the differentiation of an external (covering) cellular layer, which affords, perhaps, the nearest approach to distinct tissue-formation known among Protozoa. This envelope is formed soon after nuclear multiplication of the young trophozoite has begun, and is constituted by two nuclei and a thin, peripheral layer of cytoplasm. It remains binuclear throughout the entire period of development, and serves as a delicate cyst-membrane. The multiplication of the internal nuclei is accompanied by a corresponding division of the cytoplasm; so that instead of a multinucleate or plasmodial condition, distinct uninucleate cellules are formed, up to sixteen in number. These cellules, as a matter of fact, are sexual elements or gametes; and eight of them can be distinguished from the other eight by slight differences in the nuclei. The gametes unite in couples, each couple being most probably composed of dissimilar members: in other words, conjugation is slightly anisogamous. Each of these eight copulae gives rise to a spore.
As the name of the order implies, there are always eight spores formed. These differ from other Endosporan spores in having invariably a ternary symmetry and constitution (fig. 9). The wall of the spore is composed of three valves, each formed from an enveloping cell, and three capsular cells, placed at the upper or anterior pole, and containing each a polar-capsule, visible in the fresh condition. The valves are usually prolonged into processes or appendages, whose form and arrangement characterize the genus; but in Sphaeractinomyxon the spore is spherical and lacks processes. The sporoplasm may be either a plasmodial mass, with numerous nuclei, or may form a certain number of uninuclear sporozoites. A remarkable feature in the development of the spore is that the germinal tissue (sporoplasm) arises separate from and outside the cellules which give rise to the spore-wall; later, when the envelopes are nearly developed, the sporoplasm penetrates into the spore.
Four genera have been made known. (1) Hexactinomyxon, Stolc. Spores having the form of an anchor with six arms; sporoplasm plasmodial, situate near the anterior pole of the spore. One sp. H. psammoryctis, from Psammoryctes. (2) Triactinomyxon, St. Spores having the form of an anchor with three arms; distinct sporozoites, disposed near the anterior pole. T. ignotum, with eight spores, from Tubifex tubifex, and also from an unspecified Tubificid; another sp., unnamed, with 32 sporozoites, also from T. t. (3) Synactinomyxon, St. Spores united to one another, each having two aliform appendages; sporoplasm plasmodial. One sp., S. tubificis, from T. rivulorum. (4) Sphaeractinomyxon, C. and M. Spores spherical, without aliform prolongations; sporoplasm gives rise to very many sporozoites, occupying the whole spore. One sp., S. stolci, from Clitellio and Hemitubifex.

|
| From Lankester’s Treatise on Zoology, vol. Protozoa. |
| Fig. 9.—Spores of Actinomyxidia (after Stolc). |
|
a, Hexactinomyxon psammoryctis (par. Psammoryctes
barbatus). |

|

|
| From Wasielewski’s Sporozoenkunde. | |
|
Fig. 10.—A. Sarcosporidia in the ox; a transverse section of the oesophagus, natural size, showing the parasites in the outer (a, b, c, d, e) and inner (f, g, h) muscular coats. B. Longitudinal section of a muscle-fibre containing a Sarcosporidian parasite, × 60. |
3. Order—Sarcosporidia. With the exception of one or two forms occurring in reptiles, these parasites are always found in warm-blooded Vertebrates, usually Mammals. They are of common occurrence in domestic animals, such as pigs, sheep, horses and (sometimes) cattle. A Sarcosporidian has also been described from man. The characteristic habitat is the striped muscle, generally of the oesophagus (fig. 10, A) and heart, but in acute cases the parasites overrun the general musculature. When this occurs, as often happens in mice, the result is usually fatal. Unless, however, the organisms thus spread throughout the body, the host does not appear to suffer any serious consequences. In addition to the effects produced by the general disturbance to the tissues, the attacked animals have apparently to contend—at any rate in the case of Sarcocystis tenella in the sheep—with a poison secreted by the parasite. For Laveran and Mesnil (27) have isolated a toxine from this form, which they have termed sarcocystin.
In the early stages of growth, a Sarcosporidian appears as an elongated whitish body lodged in the substance of a muscle-fibre; this phase has long been known as a “Miescher’s tube,” or Miescheria. The youngest trophozoites that have been yet observed (by Bertram, 1) were multinucleate (fig. 11, A), but there is no reason to doubt that they begin life in a uninuclear condition. The protoplasm is limited by a delicate cuticle. With growth, organellae corresponding to the Myxosporidian pansporoblasts are formed by the segregation internally of little uninuclear spheres of protoplasm. At the same time, a thick striated envelope is developed around the parasite, which later comes to look like a fur of fine filaments. The probable explanation of this feature (given by Vuillemin, 44) is that it is due to the partial breaking down of a stiff, vertically (or radially) striated external layer (fig. 12, A), such as is seen in Myxidium lieberkühnii. Immediately internal to this is a thin, homogeneous membrane, which sends numerous partitions or septa inwards; these divide up the endoplasm into somewhat angular chambers or alveoli (fig. 12). In each chamber is a pansporoblast, which divides up to produce many spores; hence the spores formed from different pansporoblasts are kept more or less separate. The pansporoblasts originate, in a growing Sarcosporidian, at the two poles of the body, where the peripheral endoplasm with its nuclei is chiefly aggregated. More internally, spore-formation is in progress; and in the centre, pansporoblasts full of ripe spores are found.
By this time the parasite has greatly distended the muscle-fibre in which it has hitherto lain, absorbing, with its growth, practically all the contractile-substance, until it is surrounded only by the sarcolemma and sarcoplasm. It next passes into the adjacent connective-tissue, and in this phase has been distinguished from Miescheria as Balbiania, under the impression that the two forms were quite distinct. In the later stages, the parasite may become more rounded, and a cyst may be secreted around it by the host’s tissue. In these older forms, the most centrally placed spores degenerate and die, having become over-ripe and stale.

|

|
After Bertram, from Wasielewski’s | |
|
Fig. 11.—Stages in the growth of Sarcocystis tenella of the sheep. A, Youngest observed stage in which the radially striated outer coat has not appeared; the body of the trophozoite is already divided into a number of cells or pansporoblasts (k). B and C, Older stages with numerous pansporoblasts and two envelopes, an inner membrane and an outer radially striated layer. |
With regard to the spores themselves and what becomes of them, our knowledge is defective. Two kinds of reproductive germ have been described, termed respectively gymnospores (so-called sporozoites, “Rainey’s corpuscles”) and chlamydospores, or simply spores. It seems probable that the former serve for endogenous or auto-infection, and the latter for infecting fresh hosts. Unfortunately, however, both kinds of germ are not yet known in the case of any one species. The gymnospores, which are the more commonly found (e.g. in S. muris, S. miescheriana of the pig, and other forms), are small sickle-shaped or reniform bodies which are more or less amoeboid, and capable of active movement at certain temperatures. They appear to be naked, and consist of finely granular protoplasm, containing a single nucleus and one or two vacuoles. The chlamydospores, or true spores, occur in S. tenella of sheep (fig. 13), and have been described by Laveran and Mesnil (26). They also are falciform, but one extremity is rounded, the other pointed. There is a very thin, delicate membrane, most unlike a typical, resistant spore-wall; and the spores themselves are extremely fragile and easily acted upon and deformed by reagents, even by distilled water. The rounded end of the spore contains a large nucleus, while at the other end is an oval, clear space, which, in the fresh condition, shows a distinct spiral striation. The exact significance of this structure has been much debated. In position and appearance it recalls the polar-capsule of a Myxosporidian spore. The proof of this interpretation would be the expulsion of a filament on suitably stimulating the spore; while, however, some investigators have asserted that such a filament is extruded, this cannot be regarded as at all certain. Hence it is still doubtful whether this striated body really corresponds to a polar-capsule.

|
 |
| From Wasielewski’s Sporozoenkunde. | |
|
Fig. 12.—A, Sarcocystis miescheriana (Kühn) from the pig: late stage in which the body has become divided up into numerous chambers or alveoli, each containing a number of germs. | |
|
B, Sarcocystis of the ox: section of a stage similar to fig. 12. a, Substance of muscle-fibre; b, envelope of parasite; c, nuclei of the muscle; d, parasitic germs (gymnospores); e, walls of the alveoli. In the peripheral alveoli are seen immature germs. | |

|
|
(After Laveran and Mesnil, from Lankester’s Treatise on Zoology, vol. Protozoa.) |
|
Fig. 13.—Spores of Sarcocystis tenella, Raill., from the sheep. a, Spore in the fresh condition, showing a clear nucleus (n) and a striated body or capsule (c). b, Stained spore; the nucleus (n) shows a central karyosome; the striations of the polar capsule (c) are not visible. |
Nothing whatever is known as to the natural means by which infection with Sarcosporidia is brought about. Smith (39) showed that mice can be infected with Sarcocystis muris by simply feeding them on the flesh of infected mice. It is not very likely, however, that this represents the natural mode, even in the case of mice; and it certainly cannot do so in the case of Herbivora. The difficulty in the way is the delicacy of the spores, which seem totally unfitted to withstand external conditions. It may be that some alternative (intermediate) host is concerned in dispersal; but this has yet to be ascertained.
All known Sarcosporidia are included in a single genus Sarcocystis, Lank. (= Miescheria + Balbiania, Blanchard.) Some of the principal species are: S. miescheriana, from pigs; S. tenella, from sheep; S. bertrami, from horses; S. blanchardi, from Bovines; S. muris, from mice; S. platydactyli, from the gecko; and lastly, S. lindemanni, described from man.
4. Order—Haplosporidia. The Sporozoa included in this order are characterized by the general simplicity of their development, and by the undifferentiated character of their spores. The order includes a good many forms, whose arrangement and classification have been recently undertaken by Caullery and Mesnil (15), to whom, indeed, most of our knowledge relating to the Haplosporidia is due. The habitat of the parasites is sufficiently varied; Rotifers, Crustacea, Annelids and fishes furnishing most of the hosts. A recent addition to the list of Protozoa causing injury to man, a Haplosporidian, has been described by Minchin and Fantham (29d), who have termed the parasite Rhinosporidium, from its habitat in the nasal septum, where it produces pedunculate tumours.

|
| From Minchin, in Lankester’s Treatise on Zoology, vol. Protozoa. |
|
Fig. 14.—Bertramia Asperospora (Fritsch) from the body-cavity of Brachionus. × 1040. a, Young form with opaque, evenly-granulated protoplasm and few refringent granules; the nuclei (n) are small, and appear to be surrounded each by a clear space. b and c, Full-grown specimens with large nuclei and clearer protoplasm, containing numerous refringent granules (r. gr.). d and e, Morula stages, derived from b and c by division of the body into segments centred round the nuclei, each cell so formed being a spore. Between the spores a certain amount of intercellular substance or residual protoplasm is left, in which the refringent granules seem to be embedded. The morula may break up forthwith and scatter the spores, or may first round itself off and form a spherical cyst with a tough, fairly thick wall. f, Empty, slightly shrunken cyst, from which the spores have escaped. g, Free spore or youngest unicellular trophozoite. h, i, j, Commencing growth of the trophozoite, with multiplication of the nuclei, which results ultimately in forms such as a and b. |
Bertramia, a well-known parasite of the body-cavity of Rotifers, will serve very well to give a general idea of the life-cycle so far as it has yet been made out (fig. 14). The trophozoite begins life as a small, rounded uninucleate corpuscle, which as it grows, becomes multinucleate. The multinuclear body generally assumes a definite shape, often that of a sausage. Later, the protoplasm becomes segregated around each of the nuclei, giving the parasite a mulberry-like aspect; hence this stage is frequently known as a morula. The uninuclear cellules thus formed are the spores, which are ultimately liberated by the break-up of the parent body. Each is of quite simple, undifferentiated structure, possesses a large, easily-visible nucleus, and gives rise in due course to another young trophozoite. In some instances, as described by Minchin, the sporulating parasite becomes rounded off and forms a protective cyst, doubtless for the protection of the spores during dissemination.
In some forms (e.g. Haplosporidium and Rhinosporidium) the spore-mother-cells, instead of becoming each a single spore, as in Bertramia, give rise to several, four in the first case, many in the latter. Sometimes, again, the spore, while preserving the essentially simple character of the sporoplasm, may be enclosed in a spore-case; this may have the form of a little box with a lid or operculum, as in some species of Haplosporidium, or may possess a long process or tail, as in Urosporidium (fig. 15).
The Haplosporidia are divided by Caullery and Mesnil into three families, Haplosporidiidae, Bertramiidae and Coelosporidiidae; one or two genera are also included whose exact position is doubtful.
(a) Haplosporidiidae: 3 genera, Haplosporidium, type-species H. heterocirri; Urosporidium, with one sp., U. fuliginosum; all parasitic in various Annelids; and Anurosporidium, with the species A. pelseneeri, from the sporocysts of a Trematode, parasitic on Donax.

| ||
|
From Caullery and Mesnil, Archives de zoologie expérimentale, vol. 4, 1905, by permission of Schleicher Frères et Cie, Paris. | ||
| Fig. 15.—Spores of various Haplosporidia. | ||
|
1. Haplosporidium heterocirri: 2, H. scolopli. |
3, H. vejdovskii. 4, Urosporidium fuliginosum: | |
(b) Bertramiidae: 2 genera, Bertramia, with B. capitellae from an Annelid and B. asperospora, the Rotiferan parasite above described; and Ichthyosporidium, with I. gasterophilum and I. phymogenes, parasitic in various fish.
(c) Coelosporidiiae: genera Coelosporidium, type-species C. chydoriclola; and Polycaryum, type-species P. branchiopodianum. These forms are parasitic in small Crustacea. The genus Blastulidium is referred, doubtfully, by Caullery and Mesnil to this family; but certain phases of this organism seem to indicate rather a vegetable nature.
The genus Rhinosporidium should probably be placed in a distinct family. The only species so far described is R. kinealyi from the nasal septum of man, to which reference has above been made. Another form, Neurosporidium cephalodisci, agreeing in some respects with Rhinosporidium, has been described by Ridewood and Fantham (37a) from the nervous system of Cephalodiscus.
A parasite whose affinities are doubtful, but which is regarded by Caullery and Mesnil as allied to the Haplosporidia, is the curious parasite originally described by Schewiakoff as “endoparasitic tubes” of Cyclops; it has been named by Caullery and Mesnil, Scheviakovella. This organism is remarkable in one or two ways: it possesses a contractile vacuole; the amoeboid trophozoites tend to form plasmodia; and the spores, of the usual simple type, may apparently divide by binary fission.
5. There remain, lastly, certain forms, which are conveniently grouped together as “Sporozoa incertae sedis,” either for the reason that it is impossible to place them in any of the well-defined orders, or because their life-cycle is at present too insufficiently known. Serosporidia is the name given by Pfeiffer to certain minute parasites of the body-cavity of Crustacea; they include Serosporidium, Blanchardina and Botellus. Lymphosporidium, a form with distributed nucleus, causing virulent epidemics among brook-trout, is considered by Calkins (3) to be suitably placed here. Another parasite of lymphatic spaces and channels is the remarkable Lymphocystis, described by Woodcock (46), from plaice and flounders, which in some respects rather recalls a Gregarine. The group Exosporidia was founded by Perrier to include a peculiar organism, ectoparasitic on Arthropods, to which the name of Amoebidium had been given by Cienkowsky. It has recently been shown, however, that this organism is most probably an Alga. Another genus, Exosporidium, described by Sand (38), is placed at present in this group. For details of the structure of these forms and others like Siedleckia, Toxosporidium, Chitonicium Joyeuxella and Metschnikovella, a comprehensive treatise on the Sporozoa, such as that of Minchin, should be consulted.
To complete this article, it will be sufficient to mention various enigmatical bodies, associated with different diseases, which are regarded by their describers as Protozoa. Among such is the “Histosporidium carcinomatosum” of Feinberg, which he finds in cancerous growths. Cytoryctes, the name given to “Guarnieri’s bodies” in small-pox and vaccinia, has been recently investigated by Calkins (3a), who has described a complex life-cycle for the alleged parasite. Other workers, however, such as Siegel, give a quite different account of these bodies, and, moreover, find similar ones in scarlet-fever, syphilis, &c.; while yet others (e.g. Prowazek) deny that they are parasitic organisms at all.
Bibliography.—(For general works see under Sporozoa.) (1) Bertram, “Beiträge zur Kenntnis der Sarcosporidien,” Zool. Jahrb. Anat. 5, 1902; (2) L. Brasil, “Joyeuxella toxoides,” (n.g., n.sp.), Arch. zool. exp. N. et R. (3) 10, p. 5, 7 figs., 1902; (3) G. N. Calkins, “Lymphosporidium truttae,” (n.g., n.sp.), Zool. Anz. 23, p. 513, 6 figs., 1903; (3a) ib. The Life-History of Cytoryctes Variolae; Guarnieri, “Studies path. etiol. variola,” J. Med. Research (Boston, 1904), p. 136, 4 pls.; (3b) M. Caullery and A. Chappellier, “Anurosporidium pelseneeri, (n.g., n.sp.), Haplosporidie,” &c., C. R. soc. biol. 60, p. 325, 1906; (4) M. Caullery and F. Mesnil, “Sur un type nouveau” (Metchnikovella, n.g.), C. R. ac. sci. 125, p. 787, 10 figs., 1897; (5) ib. “Sur trois Sporozoaires parasites de la Capitella,” C. R. soc. biol. 49, p. 1005, 1877; (6) ib. “Sur un Sporozoaire aberrant” (Siedleckia, n.g.), op. cit. 50, p. 1093, 7 figs., 1898; (7) ib. “Sur le genre Aplosporidium” (nov.), op. cit. 51, p. 789, 1899; (8) ib. “Sur les Aplosporidies,” C. R. ac. sci. 129, p. 616, 1899; (9) ib. “Sur les parasites intimes des Annélides” (Siedleckia, Toxosporidium), C. R. ass. franç., 1899, p. 491, 1900; (10) ib. “Sur un type nouveau (Sphaeractinomyxon, n.g.) d’Actinomyxidies,” C. R. soc. biol. 56, p. 408, 1904; (11) ib. “Phénomènes de sexualité dans le développement des Actinomyxidies,” op. cit. 58, p. 889, 1905; (12) ib. “Recherches sur les Actinomyxidies,” Arch. Protistenk. 6, p. 272, pl. 15, 1905; (13) ib. “Sur quelques nouvelles Haplosporidies d’Annélides,” C. R. soc. biol. 58, p. 580, 6 figs., 1905; (14) ib. “Sur des Haplosporidies parasites de poissons marins,” ib. p. 640, 1905; (15) ib. “Recherches sur les Haplosporidies,” Arch. zool. exp. (4) 4, p. 101, pls. 11-13, 1905; (16) L. Cohn, “Über die Myxosporidien von Esox lucius,” Zool. Jahr. Anat. 9, p. 227, 2 pls., 1896; (17) ib. “Zur Kenntniss der Myxosporidien,” Centrbl. Bakt. 1, Orig. 32, p. 628, 3 figs., 1902; (18) ib. “Protozoen als Parasiten in Rotatorien,” Zool. Anz. 25, p. 497, 1902; (19) F. Doflein, “Über Myxosporidien,” Zool. Jahr. Anat. 11, p. 281, 6 pls., 1898; (20) ib. “Fortschritte auf dem Gebiete der Myxosporidienkunde,” Zool. Centrbl. 7, p. 361, 1899; (21) R. Gurley, “The Myxosporidia,” Bull. U.S. Fish. Comm., 1892, p. 65, 47 pls., 1894; (22) E. Hesse, “Sur une nouvelle Microsporidie tétrasporée du genre Gurleya,” C. R. soc. biol. 55, p. 495, 1903; (23) ib. “Thelohania légeri” (n.sp.), op. cit. 57, pp. 570-572, 10 figs., 1904; (24) ib. “Sur Myxocystis Mrazeki Hesse,” &c., op. cit. 58, p. 12, 9 figs., 1905; (25) A. Laveran and F. Mesnil, “Sur la multiplication endogène des Myxosporidies,” op. cit. 54, p. 469, 5 figs., 1902; (26) ib. “Sur la morphologie des Sarcosporidies,” op. cit. 51, p. 245, 1899; (27) ib. “De la Sarcocystin,” op. cit. p. 311, 1899; (28) L. Léger, “Sur la sporulation du Triactinomyxon,” op. cit. 56, p. 844, 4 figs., 1904; (29) ib. “Considérations sur ... les Actinomyxidies,” op. cit. p. 846, 1904; (29a) L. Léger and E. Hesse, “Sur une nouvelle Myxosporidie, Coccomyxa, n.g.,” C. R. ac. sci., 1st July 1907; (29b) ib. “Sur la structure de la paroisporale des Myxosporidies,” op. cit. 142, p. 720, 1906; (29c) A. Lutz and A. Splendore, “Über ‘Pébrine’ and verwandte Mikrosporidien,” Centrbl. Bakt. 1, 33, Orig. p. 150, 1903, and 36, Orig. p. 645, 2 pls., 1904; (29d) E. A. Minchin and H. B. Fantham, “Rhinosporidium kinealyi” (n.g., n.sp.), Q. J. Micr. Sci. 49, p. 521, 2 pls., 1905; (30) A. Mrazek, “Über eine neue Sporozoenform” (Myxocystis), S. B. Böhm. Ges. 8, 5 pp., 9 figs., 1897; (31) ib. “Glugea lophii,” Doflein, op. cit. 10, 8 pp., 1 pl., 1899; (32) C. Perez, “Sur un organisme nouveau, Blastulidium,” C. R. soc. biol. 55, p. 715, 5 figs., 1903; (33) ib. “Sur nouvelles Glugéidées,” op. cit. 58, pp. 146-151, 1905; (34) ib. “Microsporidies parasites des crabes,” Bull. sta. biol. d’Arcachon, 8, 22 pp., 14 figs., 1905; (35) W. S. Perrin, “Pleistophora periplanetae,” Q. J. Micr. Sci. 49, p. 615, 2 pls., 1906; (36) L. Plate, “Über einen einzelligen Zellparasiten” (Chitonicium), Fauna Chilensis, 2, pp. 601, pls., 1901; (37) M. Plehn, “Über die Drehkrankheit der Salmoniden” (Lentospora, n.g.), Arch. Protistenk. 5, p. 145, pl. 5, 1904; (37a) W. J. Ridewood and H. B. Fantham, “Neurosporidium cephalodisci, n.g., n.sp.,” Q. J. Micr. Sci. 51, p. 81, pl. 7, 1907; (38) R. Sand, “Exosporidium marinum” (n.g., n.sp.), Bull. soc. micr. belge, 24, p. 116, 1898; (39) T. Smith, “The production of sarcosporidiosis in the mouse,” &c., J. Exp. Med. 6, p. 1, 4 pls., 1901; (40) W. Stempell, “Über Thelohania mülleri,” Zool. Jahr. Anat. 16, p. 235, pl. 25, 1902; (41) ib. “Über Polycaryum branchiopodianum” (n.g., n.sp.), Zool. Jahrb. Syst. 15, p. 591, pl. 31, 1902; (42) ib. “Über Nosema anomalum,” Arch. Protistenk, 4, p. 1, pls. 1-3, 1904; (43) P. Thélohan, “Recherches sur les Myxosporidies,” Bull. sci. France belg. 26, p. 100, 3 pls., 1895; (44) P. Vuillemin, “Le Sarcocystis tenella, parasite de l’homme,” C. R. ac. sci. 134, p. 1152, 1902; (45) H. M. Woodcock, “On Myxosporidia in flat fish,” Proc. Liverp. Biol. Soc. 18, p. 126, pl. 2, 1904; (46) ib. “On a remarkable parasite” (Lymphocystis), op. cit. p. 143, pl. 3, 1904. (H. M. Wo.)

