towards prisoners, especially political prisoners, and by the stern measures which the government of the tsar felt compelled to adopt in order to repress the revolutionary movement. His indignation carried him into accord for a time with those who advocated the terrorist policy. In consequence he exposed himself to danger by remaining in Russia, and in 1880 he was obliged to leave the country. He settled for a short time in Switzerland, then a favourite resort of revolutionary leaders, and after a few years came to London. He was already known in England by his book, Underground Russia, which had been published in London in 1882. He followed it up with a number of other works on the condition of the Russian peasantry, on Nihilism, and on the conditions of life in Russia. His mind gradually turned from belief in the efficacy of violent measures to the acceptance of constitutional methods; and in his last book, King Stork and King Log, he spoke with approval of the efforts of politicians on the Liberal side to effect, by argument and peaceful agitation, a change in the attitude of the Russian government towards various reforms. Stepniak constantly wrote and lectured, both in Great Britain and the United States, in support of his views, and his energy, added to the interest of his personality, won him many friends. He was chiefly identified with the Socialists in England and the Social Demo- cratic parties on the Continent; but he was regarded by men of all opinions as an agitator whose motives had always been pure and disinterested. Stepniak was killed by a railway engine at a level crossing at Bedford Park, Chiswick, where he resided, on the 23rd of December 1895. He was cremated at Woking on the 28th of December. (H. H. F.)
STEPPE (from the Russ. stepi, a waste), the name given to
the level treeless plains in certain parts of the Russian Empire,
and thence sometimes, though not commonly, extended, in
physical geography, to signify similar plains elsewhere. The
name is most commonly applied specifically to the plains in
the south and south-east of European Russia and in the south-west of Asiatic Russia, and in this connexion the term sometimes
connotes semi-desert conditions. Otherwise the Russian steppes
may be considered as kindred to and connected with the Heiden
(heaths) of northern Germany.
STEPPES, GENERAL-GOVERNORSHIP OF, a portion of Russian Central Asia which includes both what was formerly known as the Kirghiz Steppe, and the region around Omsk, which was formerly part of Western Siberia. It consists of four provinces: Akmolinsk, Semipalatinsk, Turgai and Uralsk, having a total area of 711,000 sq. m. and a total population of 2,472,931 in 1897. Details are given under the names of the provinces respectively. Omsk is the capital.
STEREOBATE (Gr. στερεός, solid, and βάσις, a base), the term in architecture given to the substructure of rough masonry of
a Greek temple.
STEREOCHEMISTRY (Gr. στερεός, solid, and chemistry), a branch of chemistry which considers the spatial arrangement of the atoms composing a molecule (see Stereo-isomerism).
STEREO-ISOMERISM, or Stereomerism, a term introduced by Victor Meyer (by way of his denomination stereo-chemistry for “chemistry in space”) to denote those cases of isomerism, i.e. the difference of properties accompanying identity of molecular formulae, where we are forced to admit the same atomic linking and can only ascribe the existing difference to the different relative position of atoms in the molecule.
Historical.—Considerations concerning the relative position of atoms have been traced back as far as Swedenborg (1721); in more recent times the first proposal in this direction seems due to E. Paterno (1869), followed by Auguste Rosenstiehl and by Alexis Gaudin (1873). The step made by J. A. Le Bel and J. H. van't Hoff (1874) brought considerations of this kind in the reach of experimental test, and so led to " stereo- chemistry." The work of Louis Pasteur on molecular asymmetry in tartaric acid (i860) touched stereo-chemistry so nearly that, had structural chemistry been sufficiently developed then, stereo-chemistry might have originated fourteen years earlier; it happened, however, that Wislicenus's investigation of lactic acids (1869) immediately stimulated Van't Hoff's views. The fundamental conceptions, of Le Bel and Van't Hoff differ in that the former are based on Pasteur's notions of molecular asymmetry, the latter on structural chemistry, especially as developed by August Kekule for quadrivalent carbon. Both seem to lead to the same conclusions as to stereo-isomerism, but the latter has the advantage of allowing a more detailed insight, whereas the former, which is free from hypothetical conceptions, is of absolute reliability.
As our knowledge of stereo-isomerism originated in the chemistry of carbon compounds and found the largest development there, this part will be treated first.
Stereo-isomerism in Carbon Compounds.
1. The Asymmetric Carbon Atom.—Though stereo-chemistry is based on the notion of atoms, there is not the least danger that it may break down when newer notions about those atoms are intro- duced. Even admitting that they are of a compound nature, i.e. built up from smaller electrical particles or anything else and able to split up under given conditions, their average lapse of existencs is long enough to consider them as reliable building-stones of the molecule, though these building-stones may give way now and then, as our best ordinary ones by the action of an earthquake. Another thing which stereo-chemistry abstracts beforehand is the movement of atoms, which is generally accepted to exist, but becoming less as the temperature sinks and disappearing at absolute zero. And so the following symbols, representing atoms in a fixed position, may correspond to these last circumstances, whereas at ordinary temperatures atoms may vibrate, for instance, with these fixed positions as centres.
The first development from structural to stereo-chemistry was to consider the relative position of atoms in methane, CH4. Structural chemistry had proved that the four atoms of hydrogen were linked to carbon and not to each other, thus
| C | —H |
| —H | |
| —H | |
| —H |
and not for example
H—H·C<H
H, but how the four were grouped remained to decide.
The decision is derived as follows:—
If the four hydrogen atoms are supposed to be in a plane on one side of the carbon atom as above, two methylchlorides CH3Cl should be possible, viz. :—
| C | —Cl | and | C | —H |
| —H | —Cl | |||
| —H | —H | |||
| —H | —H |
Such isomeric compounds have never been found, but they appear as soon asthe four atoms (or groups of atoms) to which carbon is combined are different, for example in CHFClBr, fiuorchlorbrom- methane. Then and only then two isomeric compounds have been regularly observed, and the sole notion about relative position of atoms in methane which explains this fact is that the four groups combined with carbon are placed at the summits of a tetrahedron whose centre is formed by carbon. The two possibilities are then represented by:—
 |
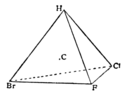 |
| Fig. 1. | Fig. 2. |
These groupings have the character of enantiomorphism, i.e. they are non-identical mirror images. If any of the two differences in the summits is given up, for example, F substituted by Cl with the formation of CHCUBr, the enantiomorphism disappears.
The isomerism corresponding to this difference in relative position is the simplest case of stereo-isomerism. The carbon atom in the special condition described, linked to four different atoms or groups, is denominated “asymmetric carbon”, and will be denoted in the following formulae as C. Stereo-isomerism exists in tartaric acid, HO2C·CH(OH)·CH(OH)·CO2H (studied by Pasteur), in the lactic acid, CH3·CH·OH·CO2H (studied by Wislicenus), while the simplest case at present known is the chlorobromofluoracetic acid, C·Cl·Br·F·CO2H, obtained by Schwartz. This stereo-isomerism, due to the presence of asymmetric carbon, is of a characteristic kind, which is in perfect accordance with the theory of its origin, being the most complete identity combined with the difference that exists between the left and right hand. All the properties which
